What is the Current Evidence on Vertebral Body Osteonecrosis?: A Systematic Review of the Literature
Article information
Abstract
Osteonecrosis (ON) is a clinical entity characterized by a pattern of cell death and complex process of bone resorption and formation. Studies related to ON have largely focused on certain anatomical sites; however, the evidence on vertebral body ON (VBON) is largely inconsistent and fragmented. The aim of this study was to clarify the pathophysiology, risk factors, imaging findings, and available treatment modalities for VBON. A systematic review of the relevant articles published in English was performed using PubMed, Embase, Medline, Google Scholar, Cochrane Central Register of Controlled Trials (CENTRAL), and CINAHL databases. A total of 81 articles were included in this study. Three main topics about VBON were identified: (1) pathophysiology and risk factors, (2) diagnosis, and (3) treatment. Forty-five studies were based on the pathophysiology, 52 on diagnosis, and 38 on the treatment options for VBON. The literature on VBON was limited and mainly focused on post-traumatic cases with a considerable overlap with nonunion and pseudoarthrosis.
Introduction
Osteonecrosis (ON) is a clinical entity characterized by a pattern of cell death and complex process of bone resorption and formation [1]. It has been reported to occur in several locations of the human body, including femoral heads, humeral heads, femoral condyles, distal end of the tibia, vertebral bodies, carpal and tarsal bones, metacarpals, and mandible [2]. Studies related to ON have largely focused on only certain anatomical sites. ON of the femoral head is a well characterized pathology which typically affects relatively young, active people aged 20–40 years. It has been estimated that 20,000–30,000 new cases of ON are diagnosed annually in the United States [3]. Medication-related ON of the jaw (ONJ) has been widely described in the recent literature. For instance, the risk of ONJ among patients treated for osteoporosis with antiresorptive and antiangiogenic medications has been reported to be approximately 0.1% [4].
Evidence on vertebral body ON (VBON) is inconsistent and fragmented and mainly limited to post-traumatic cases, with a considerable overlap with nonunion and pseudoarthrosis. The actual incidence of this entity remains unknown. An idiopathic etiology for VBON has been usually reported. The affected vertebra is usually located in the lower thoracic or upper lumbar region (T8–L4), owing to the well-known prevalence of vertebral fractures at the thoracolumbar junction. In the majority of cases, only a single vertebra is involved [5]. A correct diagnosis is essential for adequate treatment, and incorrect therapeutic management can lead to adult spinal deformity, neurological deficit, disability, and poor quality of life.
In this study, we intend to clarify the pathophysiology, risk factors, imaging findings, and available treatment modalities for VBON based on a comprehensive review of the literature.
Materials and Methods
We performed a systematic review of the available English literature on VBON using PubMed, Embase, Medline, Google Scholar, Cochrane Central Register of Controlled Trials (CENTRAL), and CINAHL databases. Combinations of the following keywords were used to retrieve the relevant articles: vertebral body ON, Kümmell’s disease (KD), intravertebral vacuum cleft (IVC), diagnosis, treatment, and outcomes. Two reviewers (MF and AZ) independently screened the titles, abstracts, and full texts (when necessary) to identify the eligible studies. Full text reports of the selected studies were included in the analysis. Reference lists of the identified articles were manually screened to include all relevant articles. Duplicate publications, review articles, experts’ comments, and articles not published in English language were excluded. The included papers were categorized into three groups according to the main topic: (1) pathophysiology and risk factors, (2) diagnosis, and (3) treatment. The level of evidence of a given study was assigned based on the 2005 scoring system adopted by the North American Spine Society [6]. The PRISMA (preferred reporting items for systematic review and meta-analysis) 2009 checklist was followed while performing this review.
Results
A total of 408 articles were initially retrieved from the databases, of which 85 duplicates were excluded. Finally, 81 articles related to VBON were included in the review: 11 were level III studies, 39 were level IV, and 31 were level II. The article typologies considered for this study included case reports, case series, and retrospective and prospective studies on VBON. Forty-five studies focused on the pathophysiology and risk factors, 52 on the diagnosis, and 38 on the treatment options for VBON. A schematic illustration of the literature search and the study selection criteria are presented in Fig. 1.
Discussion
1. Pathophysiology and risk factors
The most substantial evidence available regarding ON was with respect to the femoral head; a similar pathological pathway was presumed for ON at other anatomical sites, such as vertebral bodies. There is a general consensus in the literature about the pathogenesis and histopathology of ON; however, there is no consensus regarding its risk factors. Unfortunately, most reports were anecdotal or employed case-control methods with low level of evidence. Many clinical conditions have been associated with VBON, and several factors may act in combination. Vertebral body necrosis can be caused due to (1) cytotoxicity, (2) genetic factors, and (3) decreased intraosseous blood flow (i.e., ischemia, also known as avascular necrosis [AVN]).
1) Cytotoxicity
Direct toxic effects on the bone have been described in the literature; e.g., in patients with solid tumors treated with cytotoxic chemotherapy or those with human immunodeficiency virus infection treated with highly active antiretroviral therapy [7,8].
2) Genetic factors
Some authors hypothesized a protective or predisposing influence of certain genetic factors in the causation of femoral head AVN [2,9,10]. However, no such evidence was available for VBON.
3) Ischemia/avascular necrosis
Ischemia may be related to (1) blood vessel disruption, (2) intraluminal obliteration, or (3) extraluminal obliteration caused due to increased interstitial pressure.
Anatomical studies demonstrated that the posterior two-thirds of vertebral bodies receive collateral blood from four arteries derived from two intervertebral levels, whereas the ventral side does not. Hence, the anterior one-third of the vertebral body is defined by some authors as the “watershed zone” associated with a higher risk of ischemic AVN [11-14].
Post-traumatic vascular disruption is one of the most frequent mechanisms for the development of ON. The possible pathogenesis is based on the interruption or reduction of blood supply consequent to fracture and inadequate revascularization of the bone marrow [2]. Nevertheless, there are several notable points. In the literature, trauma and time interval between trauma and ON development have not been clearly defined. Moreover, there is a considerable overlap between various reported clinical entities, such as major or minor trauma in the osteopenic or osteoporotic bone and nonunion caused by instability or vascular damage at the fracture site [5,8,15-28].
In this intricate condition, KD is also worth mentioning. KD is a clinical condition in which patients develop a painful progressive angular kyphosis as a result of delayed vertebral body collapse (VBC) after a minor spinal trauma [5,24,29-32]. Kü mmell theorized this clinical condition before the advent of radiography [33,34]. Only after X-ray examinations were routinely available was KD recognized as a distinct clinical entity. Steel [33] and Ringler [34] clearly demonstrated that the kyphosis was a consequence of delayed VBC in the absence of radiological evidence of vertebral disruption immediately after trauma [32]. Ideally, KD is diagnosed on the basis of repeated X-ray examinations, wherein the earliest films do not show any signs of fracture [29,31,32]. The actual incidence of the disease is essentially unknown. Currently, AVN is believed to be the main cause of delayed post-traumatic VBC [11].
Interruption of the vascular flow in the vertebral body may result from intravascular obstruction. A variety of conditions may cause intraluminal obliteration. For instance, sickle cell crisis, Caisson disease, and prothrombosis have all been shown to cause VBON [11,35]. Pancreatitis is another risk factor for VBON. High blood levels of lipolytic enzymes cause intraosseous fat necrosis and obstruction of bone vessels by fat droplets. Another proposed mechanism is the release of pancreatic enzymes in the abdomen from a cyst [36,37]. Furthermore, arterial abnormalities, dyslipidemia (hypertriglyceridemia), leukemia, and lymphoma have been shown to be associated with a higher risk of ON [1,2,38,39].
Another mechanism implicated in VBON is extraluminal obliteration of blood vessels caused by increased interstitial pressure. Chronic glucocorticoid therapy and alcohol intake, resulting in fat embolism promotion, lipid deposition, and adipocyte hypertrophy, are also important risk factors reported in the literature [1,2,38-41]. Glucocorticoid therapy is used in a variety of diseases; therefore, it is difficult to discern the contribution of corticosteroids from that of the underlying condition. In Gaucher disease type 1, the accumulation of glucocerebrosides in histiocytic lysosomes is known to cause increased interstitial pressure [42,43]. Furthermore, intramedullary hemorrhage due to trauma or vascular disruption may lead to increased marrow pressure, which in turn may predispose an individual to AVN [44].
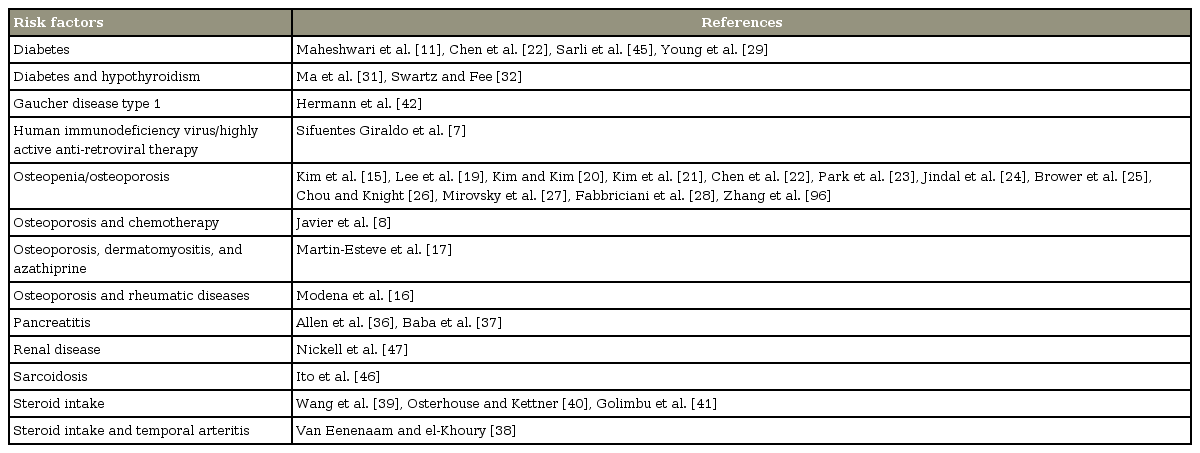
Other implicated risk factors for ON have also been reported in the literature but without clear etiopathogenetic mechanism. Diabetes mellitus is a widely reported risk factor for AVN [11,29,31,32,45], probably due to metabolic osteopenia. Ito et al. [46] described the occurrence of ON in a patient with sarcoidosis. Other predisposing factors include cirrhosis, hyperuricemia, infection, and malignancy [1,47]. Table 1 summarizes the risk factors for VBON reported in the literature.
2. Diagnosis
Vertebral biopsy is the gold standard for the diagnosis of ON. Considering the invasiveness of this procedure, imaging should be as accurate as possible. Several imagingbased classification systems for femoral head ON have been developed to facilitate prognostic assessment, treatment planning, and outcome evaluation. These include Ficat-Arlet X-ray classification, Steinberg’s system, Association Research Circulation Osseous system, and Japanese Orthopaedic Association system, including computed tomography (CT) and magnetic resonance imaging (MRI) findings. Another staging classification developed by Mitchell et al. [48] relies only on MRI features to estimate the lesion severity. For VBON, no classification system has been widely accepted, and reports about imaging features are generally supported by a low level of evidence.
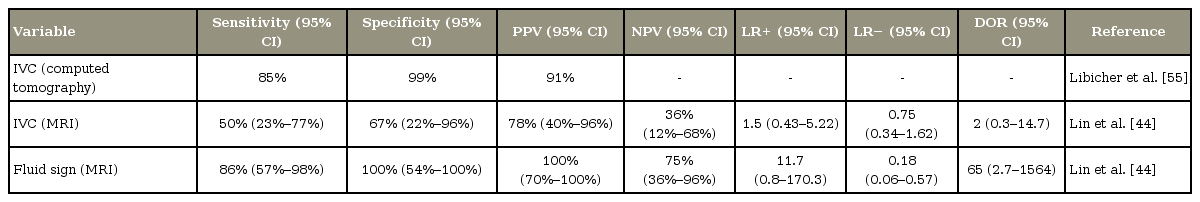
Maldague et al. [49] described a gaseous collection in the collapsed vertebral body on radiographs. This feature was referred to as IVC, and the authors considered this as a pathognomonic sign of VBON. The cleft can be detected on radiographs as horizontal accumulation of gas (95% nitrogen, oxygen, and carbon dioxide) and appears as a transverse, linear, or semilunar radiolucent shadow on plain radiographs. In some cases, it appears in extension stress and disappears in flexion [45,50]. Some authors suggested that this finding is the result of nonunion due to ischemia and instability after vertebral compression fracture. Other reports have described IVC in the setting of malignancy and infection [27]. Bhalla and Reinus [51] found that linear morphology of vacuum clefts are mostly associated with a benign process and are indicative of ischemic necrosis of the vertebral body. Several synonyms have been used in the literature to describe this feature: “linear intravertebral vacuum” [51], “intravertebral cleft” [52], “intravertebral vacuum phenomenon” [53], “intravertebral vacuum sign” [41], “Kü mmell sign” [25], and “intraosseous vacuum phenomenon” [54]. This sign is more easily detected on CT and appears more heterogeneous and irregular than that on X-ray radiographs [5,29,31,55]. Libicher et al. [56] investigated the prevalence of IVC in vertebral compression fractures using histological evaluation of biopsy specimens from the affected vertebrae. They demonstrated that IVC is a specific indicator of ON in patients with vertebral compression fractures (sensitivity, 85%; specificity, 99%; positive predictive value, 91%) and indicated local bone ischemia associated with a nonhealing vertebral collapse and pseudoarthrosis [55]. Others have also supported this theory [52,53,57-59]. Another less widely accepted theory hypothesized an intradiscal origin of gas collection in the vertebral body [60,61]. However, a retrospective study by Yu et al. [62] suggested that intradiscal gases may originate from the intravertebral body and not vice versa.
Bone scan is among the more sensitive imaging tools for the diagnosis of early ischemic necrosis; however, it is not contemplated as a routine investigation for the diagnostic workup of ON. Van Eenenaam and el-K houry [38] reported a patient who exhibited increased uptake on bone scintigraphy with normal CT and progressed to VBC in 9 weeks. Contrarily, a recent article described a cold defect on bone scan in a vertebral body with IVC sign [63].
On MRI, IVC generally appears as a hypointense area on T1- and T2-weighted images and exhibits a lack of enhancement on enhanced T1-weighted images. Collection of intravertebral fluid is also described in VBON. This MRI finding is referred as the fluid sign. The fluid collection appears as a well-defined area of low signal intensity on T1-weighted images, high signal intensity on T2-weighted images, and no enhancement on enhanced T1-weighted images [5,15,24,31,32,44,54,62-69]. Another MRI finding that is considered highly indicative of AVN is the so-called “double-line sign” (a central zone of hyperintensity surrounded by a hypointense band on T2-weighted images) [11,29,31,32,70,71], which has also been described in cases of AVN of the femoral head. This finding is believed to represent the sclerotic tissue surrounding the central new granulation tissue. Some authors have noted that progressive changes in the content of the cleft and consequently in the imaging findings may arise depending on the patients’ position [68,72]. Malghem et al. [72] reported that initially, the cleft shows an air pattern during extension of the spine, with a radiolucent band on radiographs and void signal on MRI. Later, the vacuum disappears on radiographs, and a fluid sign appears on T2-weighted images. Furthermore, Yu et al. [62] reported that the occurrence of air and fluid was approximately equal in VBON. The coexistence of air and fluid in the same affected vertebral body is not rare (21.5%). They also observed that vertebral collapse was significantly more severe in vertebral bodies that had only intravertebral air than in those that had intravertebral fluid with or without air. Additionally, they suggested that intravertebral air represents an advanced stage of the disease, whereas fluid sign may represent an earlier stage [62]; this hypothesis has been supported by Lin et al. [44] in a recent article. They also speculated that patients with a fluid sign are more prone to developing bone regeneration than those with no fluid sign and that fluid sign is a stronger indicator of ON than intravertebral vacuum phenomenon (diagnostic odds ratio, 65 versus 2; sensitivity, 86% versus 50%; specificity, 100% versus 67%) [44]. Conversely, in a retrospective comparative study by Niu et al. [73], vertebral height restoration and kyphotic deformity correction in patients with intravertebral air was significantly better than that in patients with fluid sign. Table 2 describes the accuracy of imaging findings reported in the literature.
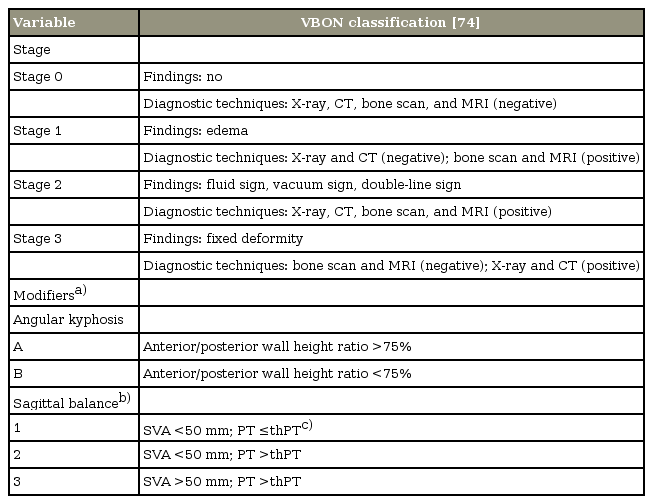
Considering the lack of a classification system and based on the evidence drawn from our review about the diagnosis of VBON, we propose a classification based on the imaging findings and sagittal parameters that may provide a basis for staging and treatment of VBON [74] (Table 3).
3. Treatment
Several factors must be considered when defining the most suitable treatment modality for VBON, including comorbidity, levels of pain and disability, time from onset, spinopelvic balance, and possible neurological involvement. Sagittal balance has been increasingly considered in the last decade. Sagittal plane alignment is closely associated with disability and quality of life, whereas coronal imbalance has a minor impact on clinical outcomes. An adequate radiological evaluation with posteroanterior and lateral full-length radiographs is mandatory prior to deciding the treatment option (surgical versus conservative approach). The vertebral surgeon must evaluate the pelvic parameters (pelvic incidence [PI], pelvic tilt [PT], and sacral slope) and sagittal alignment (lumbar lordosis [LL], thoracolumbar kyphosis [TLKKK], thoracic kyphosis [TKKK], and sacral vertical axis [SVA]). PI is the key parameter for the evaluation of global spinopelvic balance; using PI, it is possible to calculate the theoretical values of PT, LL, and TK for each patient. General alignment goals have also been established: SVA <50 mm, PT ≤theoretical PT (thPT=0.37×PI−7), TLK=0°, and LL angle >10° of PI [75]. The most common sagittal anomalies included hypolordosis or kyphosis in the lumbar spine, kyphosis in the thoracolumbar junction, and hyperkyphosis in the thoracic spine.
Sagittal alignment can be classified into balanced (SVA <50 mm and PT ≤thPT), compensating (hidden imbalance, SVA<50 mm and PT >thPT), and imbalanced (SVA >50 mm and PT >thPT) profiles. Lamartina and Berjano [76] recently proposed a classification for sagittal imbalance based on sagittal alignment and compensatory mechanism [75,77]. As far as VBON treatment is concerned, a spectrum of options have been described in the literature (Table 4).
4. Conservative management
Early reports in the literature focused on conservative treatments, such as bracing and bed rest, whereas more recent ones favored surgical intervention [33,38]. Adjuvant therapeutic tools, such as bisphosphonates, teriparatide, and capacitive coupling electrical stimulation, have been proposed in the literature [28,78].
5. Vertebral augmentation techniques
Percutaneous vertebral augmentation is a widely performed technique (vertebroplasty, kyphoplasty, and craniocaudal expandable device with consecutive bone cement injection) [15,18,71,79-87] aimed at pain relief, restoration of the vertebral body height, and correction of the deformity (Figs. 2–4). Zhang et al. [88] reported similar clinical and radiological outcomes of kyphoplasty and vertebroplasty. Vertebroplasty is less expensive, whereas kyphoplasty is associated with a lower risk of cement leakage [88]. Patients with IVC achieved better kyphosis angle reduction with percutaneous vertebral augmentation than those without IVC [89]. Various studies that examined the correlation between the presence of IVC and cement leakage have yielded conflicting results. Krauss et al. [89] reported a lower incidence in cases with IVC, whereas others identified IVC as an additional strong risk factor for cement leakage [90,91]. Tanigawa et al. [92], Wu et al. [93], and Jung et al. [94] found no statistically significant association between IVC and cement leakage. Heo et al. [95] observed that after vertebroplasty, the compression and kyphosis of VBON continued to progress for ≥2 years. As vertebroplasty may not provide sufficient stability, they strongly recommended strict observation and follow-up. It is also possible to use a combination of pedicle screw fixation in the adjacent levels and percutaneous vertebral augmentation at the necrotic level to improve stability [19,20,22,23,96].
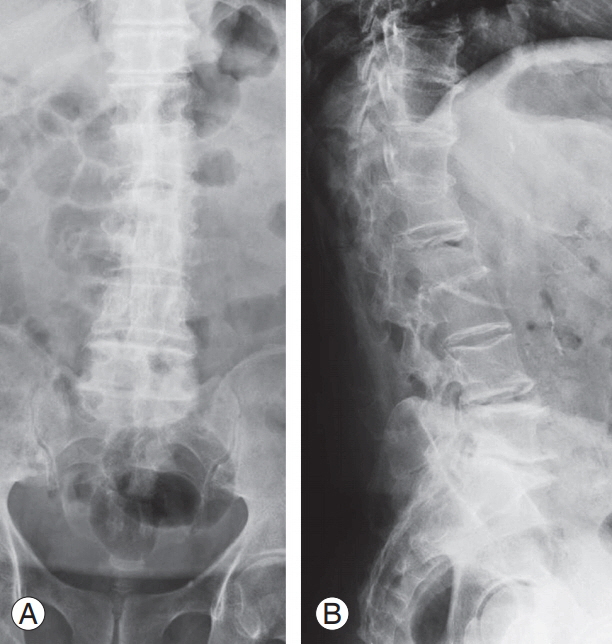
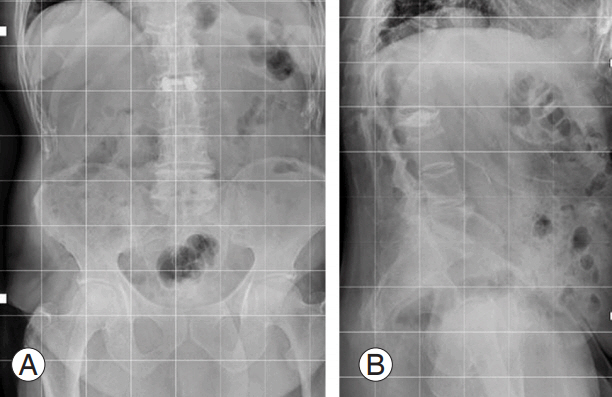
(A, B) X-ray image of an 81-year-old male patient after 5 months of brace treatment for A1 ASIA (American Spinal Injury As- American Spinal Injury As American Spinal Injury Association) E L2 vertebral fracture staged as 2, A, 3 according to our classification. The image shows intravertebral vacuum cleft compatible with vertebral body osteonecrosis. The patient suffered from uncontrolled back pain with significantly impaired quality of life.

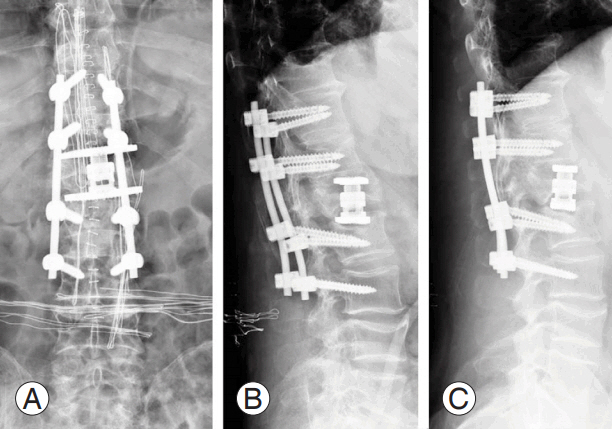
(A–C) Intraoperative images during vertebral augmentation. For high perioperative risk (American Society of Anesthesiologists 5), the surgeon opted for a bilateral transpedicular percutaneous craniocaudal expandable device to restore the original vertebral shape and bone cement injection to stabilize the reduction.
6. Major surgical management
The amount of necessary sagittal correction significantly determines the choice of surgical strategy. Surgeons must provide an adequate frame to the anterior column and sufficient instrumentation to the posterior column to support the correction until a solid fusion occurs. Surgical approach, either in one or two stages, can be performed only with a posterior, anterior, or lateral approach as well as with a combined anterior–posterior and lateral–posterior approach. However, some authors recommended a purely posterior approach in high-risk patients, such as the elderly and those with comorbidities, to avoid potential complications associated with the violation of the thoracic cavity or retroperitoneal space.
Osteoporosis is the most common comorbidity in patients with VBON, and a high rate of implant failure has been reported due to decreased bone strength and density. Augmentation methods to enhance pedicle screw fixation have improved, including instrumentation at multiple levels, bioactive cement augmentation, use of fenestrated or expandable pedicle screws [20], and sublaminar clamps and hooks to improve the stability of fusion. Posterior transpedicular vertebral bone grafting with pedicle screw fixation is another treatment option reported in the literature, although with contrasting results [39,54,97,98]. In most cases, prompt diagnosis of vertebral fracture can help prevent VBON, fix the deformity, and preclude the need for radical correction methods [30,99,100].
The most common osteotomies for the correction of sagittal imbalance are Smith-Petersen osteotomy in the lumbar spine, Ponte’s osteotomy in the thoracic spine, pedicle subtraction osteotomy (PSO), asymmetrical PSO in the presence of coronal imbalance, and Corner osteotomy [101,102]. In case of notable insufficiency of the anterior column, vertebral column resection (VCR) with the use of an expansible cage is recommended [102] (Figs. 5–7). However, these surgical techniques are associated with high rates of complications. In a recent review, the authors reported a total incidence of complications of 66% for PSO, 35% for VCR, and 45% for non-three column osteotomy [103-106].
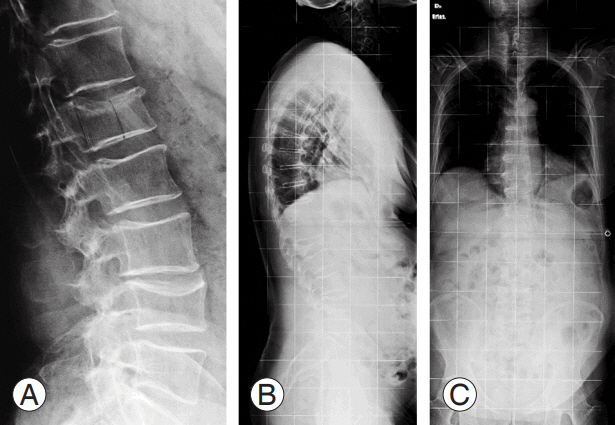
(A) X-ray image at emergency department admission of a 70-year-old male misdiagnosed with A3 ASIA (American Spinal In- ASIA (American Spinal In- (American Spinal In- American Spinal In American Spinal Injury Association) E L1 vertebral fracture. (B, C) X-ray images after 2 months of bracing treatment.
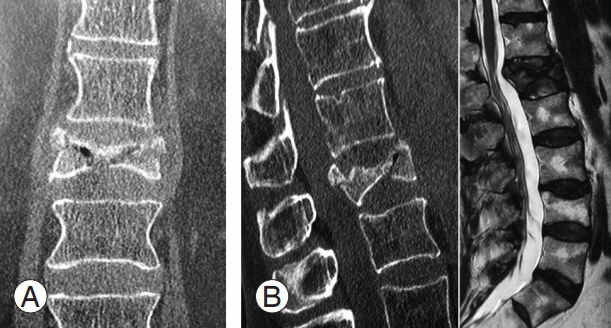
(A, B) Computed tomography at 2 months after trauma con omputed tomography at 2 months after trauma confirmed severe L1 vertebral body collapse (type A3) with vertebral body osteonecrosis and intravertebral vacuum cleft sign. (C) Magnetic resonance imaging T2-weighted image shows hypointense lesion at L1 vertebral body
Conclusions
To the best of our knowledge, this is the first systematic review that summarizes the available evidence related to the pathophysiology, risk factors, imaging findings, and available treatments for VBON.
The number of studies on VBON is limited, somewhat inconsistent, often limited to certain anatomical sites, and mainly focused on post-traumatic cases, with a considerable overlap with nonunion and pseudoarthrosis. Considering this limitation, the main reported risk factors include osteopenia/osteoporosis and steroid therapy; however, several factors can act in combination. The lack of high-grade evidence makes it difficult to clearly identify the optimal diagnostic protocol and treatment guidelines for patient management. The dualism between air and fluid clefts remains unclear and controversial. The present study suggested that further high quality research and treatment-oriented classification are needed to better understand and treat this common but unclear clinical entity.
Notes
No potential conflict of interest relevant to this article was reported.
Acknowledgements
We gratefully acknowledge H2O S.R.L. for the English version of this manuscript.