|
 |
- Search
| Asian Spine J > Volume 10(2); 2016 > Article |
Abstract
Purpose
To report the outcomes of patients with lumbar spinal stenosis treated with percutaneous endoscopic decompression, focusing on the results of clinical evaluations.
Overview of Literature
There are no studies about two portal percutaneous endoscopic decompression in the treatment of lumbar spinal stenosis.
Methods
Medical and surgical complications were examined and clinical results were analyzed for 30 patients who consecutively underwent two portal percutaneous endoscopic decompression for lumbar spinal stenosis were reviewed. The operations were performed by unilateral laminotomy for bilateral decompression.
Results
All patients displayed clinical improvement when were evaluated with visual analog scale (VAS) score of pain, Oswestry disability index (ODI) and Macnab criteria. The improvement of VAS and ODI was 8.3±0.7 to 2.3±2.6 and 65.2±13.7 to 24.0±15.5, respectively (both p<0.05). Complications were the same as for open decompression. The most common complication was transient nerve root paresthesia.
Conclusions
Surgical decompression with two portal percutaneous endoscopic decompression has initial benefits, but long-term studies should pay more attention to the risks of postoperative instability and restenosis as well as the need for re-operation. Further investigations with long-term results are thus required.
The treatment of lumbar spinal stenosis continues to increase, especially in older people. The aim of lumbar spinal canal stenosis surgery is relief of sciatic leg pain and improved walking ability by decompression of the spinal canal. The classic operation involves the sequential dissection of the paraspinal muscles and spinous process, followed by removal of the interspinous ligaments [123].
Scarring of the epidural space was problematic [456789]. Scars may become clinically symptomatic [567], which can make subsequent surgery more difficult due to the postoperative adhesion between the epidural space and paravertebral muscle [10111213]. The resection of structures preserving stability may promote operation-induced segmental instability [1314]. Destruction and denervation of the dorsal branch of the spinal nerves may be detrimental to the stabilization and coordination systems [81516]. Resection of joint and soft-tissue structures in the lateral and ventral area is also required for decompression of degenerative lumbar spinal stenosis, and this occurs often.
Minimally invasive surgery that better spares tissue are being increasingly used [1017]. New surgical techniques of disc herniations in the lumbar spine now permit a full-endoscopic procedure under continuous irrigation, and is minimally invasive [1819]. One key issue is the possibility of adequate bone and ligamentum flavum resection under continuous visual control [181920]. This also enables the technique to be used in spinal stenosis surgery [20].
We use two portal percutaneous endoscopic decompression for lumbar spinal canal stenosis. This procedure has a number of advantages. It permits the use of ordinary arthroscopic and spine instruments without need for special endoscopic sets. It allows free movement and angulation of the surgical tool and the endoscope independent of each other as they are not restricted by the confines of a common working portal which results in marked reduction in the technical difficulties. Finally, the use of saline irrigation abolishes the problem of repeated cleaning of the endoscopic lens of accumulated fog or blood. This technique is useful for the minimally invasive surgical treatment of spinal canal stenosis. However, studies of this technique have been limited.
The objective of the current study was to report the outcomes of patients with lumbar spinal stenosis treated percutaneous endoscopic decompression, focusing on the surgical techniques and the results of the patients' clinical evaluations.
This study was approved by our Institutional Review Board and informed consent was obtained from each patient. Thirty consecutive patients undergoing percutaneous endoscopic decompression for lumbar spinal stenosis between January 2012 and June 2013 were enrolled. The inclusion criteria were neurogenic claudication or radicular leg pain with associated neurologic signs referring to the lumbar spinal stenosis syndrome; moderate-to-severe spinal canal stenosis shown on cross-sectional imaging, such as magnetic resonance imaging (MRI) or computed tomography (CT) scan, and failure of conservative treatment for at least 3 months. The exclusion criteria were spondylolisthesis more than Meyerding Grade I; prior surgery in the same segment; coronal plane deformity with Cobb angle more than 10 degrees and spinal instability in dynamic radiograph. The patients were positioned prone in a kneeling position after general anesthesia. The operated level was identified with image intensifier. A spinal needle was inserted and localization was rechecked with a lateral view. Endoscope portal and working portal were inserted through the two separate skin incisions and docked onto the lamina (Fig. 1). The localization was reconfirmed with lateral view of fluoroscopy before the decompression. Potential space was created with endoscopic cautery (Fig. 2). Unilateral laminectomy was performed for patients with unilateral neurological symptoms. For patients with central spinal canal stenosis symptoms, we performed unilateral approach for bilateral decompression as described by Guiot et al. [21] to decompress bilateral lateral recesses and the central canal. The ligamentum flavum was completely excised. Decompression of the lateral recess was achieved by partial facetectomy. To preserve the integrity of the facet joint as much as possible, we used high-speed pneumatic burrs with diamond heads and curved Kerrison rongeur to undercut the facet joint. Then we moved the endoscope to the central canal and contralateral lateral recess. This process was performed by gently moving over the dural sac, excising the ligamentum flavum with straight and curved Kerrison punches and undercutting the lamina with a high-speed pneumatic burr. The adequacy of decompression was determined by probing the traversing nerve roots to confirm the extent of decompression. The endpoint of decompression was the outer edges of the bilateral nerve roots. Illustrative cases are depicted in Figs. 3, 4, 5, 6. After hemostasis, no drain was placed and the incision was closed. Ambulation was allowed immediately after the surgery with a brace. Rehabilitation was started from the day after the operation. All patients were discharged from the hospital within several days of the surgery.
Preoperative and follow-up patient functions were evaluated using the Oswestry disability index (ODI), visual analogue scale (VAS) score of pain and Macnab criteria. Neurologic claudication was recorded as walking ability (<400 m, 400–1600 m, >1600 m).The results obtained were recorded in patient charts and were not the basis for any decision making. The data about the preoperative comorbidities, intraoperative and postoperative complications were retrieved from the medical chart review.
Continuous variables are presented as means±standard deviations. Categorical variables are presented as counts and percentages. Repeated analysis of variance was performed to compare the differences at four time points of ODI and VAS. The statistical analyses were performed with SPSS software ver. 15 (SPSS Inc., Chicago, IL, USA) and two-tailed p<0.05 indicated statistical significance.
The patients ranged from 50 to 67 years of age, with a mean age of 56±6.2 years. Females predominated, with 19 women (63%) and 11 men (37%). Twenty-two patients (73%) had a concomitant health problem that included cardiac diseases (n=12, 40%), pulmonary problems (n=5, 17%) and diabetes (n=5, 17%). Symptoms were unilateral or bilateral sciatica (60%), neurogenic claudication (70%) and back pain (30%) although it was never an isolated symptom. The patients had symptoms of lumbar stenosis for a mean length of 15 months. A one level procedure was performed in 25 cases. The others were two level procedures. Typically, the side chosen for the surgical approach was the clinically more symptomatic side. This resulted in 10 patients (33%) being operated on from the right side and 20 (67%) from the left. The anatomic distribution of the operated levels showed a predominance of procedures performed at the L4–5 level in 21 patients (70%). All patients were followed until the end of the study.
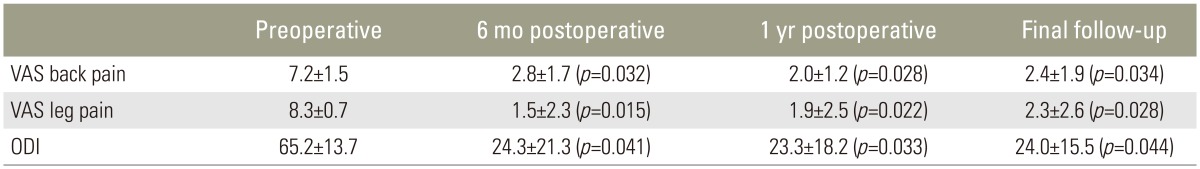
As with any new surgical technique, our proficiency with the two portal percutaneous endoscopic decompression technique improved as our experience grew. The operative time for a single-level procedure ranged from 90 to 120 minutes, with an average of 98.3±14.3 minutes and 170.4±16.1 minutes for the double-level procedure. No patient received a blood transfusion. The average length of hospitalization was 3.16±1.3 days. The duration of follow-up was at least 2 years with a range of 24–36 months. All preoperative and postoperative data were available for outcome analysis. The mean preoperative back pain VAS score was 7.2±1.5; it was 2.8±1.7 at 6 months after surgery, 2.0±1.2 after 1 year, and 2.4±1.9 at the final review (p<0.05). The mean preoperative leg pain VAS score was 8.3±0.7; it was 1.5±2.3 six months postoperatively, 1.9±2.5 at 1 year, and 2.3±2.6 at the final review (p<0.05). The mean preoperative ODI score was 65.2±13.7, at 6 months after surgery it was 24.3±21.3, 1 year after it was 23.3±18.2, and at the final review was 24.0±15.5) (p<0.05) (Table 1).
Of the patients admitted with neurogenic claudication in the preoperative study, 85% reported improvement in the ability to walk at the end of the study. Preoperatively, 90% (n=27) were able to walk painlessly for less than 400 m and 10% (n=3) for more than 400 m but less than 1,200 m. Six months after surgery, two patients (6.7%) were able to walk less than 400 m and four patients (13.3%) were able to walk more than 400 m but less than 1,200 m. At 1 year, two patients (6.6%) were able to walk less than 400 m and five patients (16.6%) were able to walk more than 400 m but less than 1,200 m. At the final review, one patient (3.3%) was able to walk less than 400 m, five patients (16.7%) were able to walk more than 400 m but less than 1,200 m and 23 patients (76.7%) were able to walk more than 1,200 m (Table 2).
According to the Macnab criteria, good results were obtained for 83% (n=25) patients, fair results for 13% (n=4) and poor result in one patient.
There were no perioperative deaths. We observed two surgery related complications (6.6%). All were transient paresthesia of the nerve root. No incidental durotomy, postoperative epidural hematoma and superficial infection were noted. During the follow-up period, one patient (3%) underwent subsequent lumbar surgery. Reoperations consisted of open spinal decompression and fusion to treat clinical relapse.
Advances in the management of lumbar spinal stenosis are being driven by the increasing number of patients requiring surgical treatment [222324]. The microendoscopic decompression technique for lumbar disorders involves bilateral decompression surgery using the unilateral approach. The procedure has been gradually and successfully applied to the treatment of lumbar spinal stenosis [2526]. We used the same technique for two portal percutaneous endoscopic decompression for lumbar spinal stenosis. This procedure has three advantages. First, ordinary arthroscopic and spine instruments are used, without the need of special endoscopic sets. Second is the free movement and angulation of the surgical tool and the endoscope independent of each other as they are not restricted by the confines of a common working portal, which results in marked reduction in technical difficulties. Third, the use of saline irrigation abolishes the problem of repeated cleaning of the endoscopic lens to remove accumulated fog or blood.
However, the surgical procedure has not yet been established internationally. One of the main barriers is the endoscopic approach to the spine. The spine is not a hollow space like the knee or shoulder, so arthroscopic visualization of the working instrument and target tissues/bone may not be easy, even with water pumping and irrigation. A potential space needs to be created before laminectomy (Fig. 2). Another hindrance is that the surgical technique involves partial resection of the bilateral lamina and total removal of the ligamentum flavum from the base of the spinous process using a pneumatic burr and rongeur. The procedure decreases perioperative complications and increases postoperative stability. Moreover, it is possible to completely preserve the contralateral facet joint. Previous investigation on facet joint resection rate after microendoscopic decompression surgery for lumbar spinal stenosis found that about 70% of the facet joints were preserved in the side of the approach and more than 95% were preserved in the contralateral side. However, maximal preservation of the facet joints is important for preventing the progression of spinal instability after surgery [1427].
In the present study, the most commonly encountered surgery-related complication was transient paresthesia of the nerve root, which has a previously reported incidence of 10% in open laminectomy for lumbar spinal stenosis. However, a learning curve is a reality of the technique concerning the surgical approach and severity of stenosis; most cases occurred during the early development of the technique, in patients undergoing unilateral laminotomy for bilateral decompression or in patients with severe stenosis. As shown in this study, percutaneous endoscopy results in an excellent viewing angle with good contralateral visualization. Hence, even in patients with severe stenosis, a dorsal space was first made by the bony procedure, so intra-operative positive pressure irrigation and the spinal decompression could be carried out more easily. Dural tearing does not occur, since the ligamentum flavum is kept as protective barrier for the dura mater until completion of the bony procedure. Manipulation of the neural tissues and extensive surgical skill of the surgeon may alleviate the risk of such complications.
There are some limitations associated with the current study. First, this study is retrospective. Second, the follow-up period was not long enough to show the long-term outcome. Because the initial benefits of surgical decompression may decline over time, additional longer-term studies should pay more attention to the risks of postoperative instability and restenosis as well as the need for re-operation.
Surgical decompression with two portal percutaneous endoscopic decompression has initial benefits, but long-term studies should pay more attention to the risks of postoperative instability and restenosis as well as the need for re-operation. Further investigations with long-term results are thus required.
Conflict of Interest
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
1. Herno A, Airaksinen O, Saari T. Long-term results of surgical treatment of lumbar spinal stenosis. Spine (Phila Pa 1976) 1993 18:1471–1474. PMID: 8235818.


2. Iguchi T, Kurihara A, Nakayama J, Sato K, Kurosaka M, Yamasaki K. Minimum 10-year outcome of decompressive laminectomy for degenerative lumbar spinal stenosis. Spine (Phila Pa 1976) 2000 25:1754–1759. PMID: 10888941.


3. Johnsson KE, Willner S, Johnsson K. Postoperative instability after decompression for lumbar spinal stenosis. Spine (Phila Pa 1976) 1986 11:107–110. PMID: 3704799.


4. Annertz M, Jonsson B, Stromqvist B, Holtas S. No relationship between epidural fibrosis and sciatica in the lumbar postdiscectomy syndrome A study with contrast-enhanced magnetic resonance imaging in symptomatic and asymptomatic patients. Spine (Phila Pa 1976) 1995 20:449–453. PMID: 7747228.


5. Fritsch EW, Heisel J, Rupp S. The failed back surgery syndrome: reasons, intraoperative findings, and long-term results: a report of 182 operative treatments. Spine (Phila Pa 1976) 1996 21:626–633. PMID: 8852320.


6. Kraemer J. Intervertebral disk diseases: causes, diagnosis, treatment and prophylaxis. Stuttgart: Thieme; 1990.
7. Schoeggl A, Maier H, Saringer W, Reddy M, Matula C. Outcome after chronic sciatica as the only reason for lumbar microdiscectomy. J Spinal Disord Tech 2002 15:415–419. PMID: 12394667.


8. Lewis PJ, Weir BK, Broad RW, Grace MG. Long-term prospective study of lumbosacral discectomy. J Neurosurg 1987 67:49–53. PMID: 3598671.


9. Ruetten S, Meyer O, Godolias G. Epiduroscopic diagnosis and treatment of epidural adhesions in chronic back pain syndrome of patients with previous surgical treatment: first results of 31 interventions. Z Orthop Ihre Grenzgeb 2002 140:171–175. PMID: 12029589.


10. Ragab AA, Fye MA, Bohlman HH. Surgery of the lumbar spine for spinal stenosis in 118 patients 70 years of age or older. Spine (Phila Pa 1976) 2003 28:348–353. PMID: 12590208.


11. Katz JN, Lipson SJ, Larson MG, McInnes JM, Fossel AH, Liang MH. The outcome of decompressive laminectomy for degenerative lumbar stenosis. J Bone Joint Surg Am 1991 73:809–816. PMID: 2071616.


12. LaRocca H, Macnab I. The laminectomy membrane: studies in its evolution, characteristics, effects and prophylaxis in dogs. J Bone Joint Surg Br 1974 56:545–550. PMID: 4421702.

13. Markwalder TM. Surgical management of neurogenic claudication in 100 patients with lumbar spinal stenosis due to degenerative spondylolisthesis. Acta Neurochir (Wien) 1993 120:136–142. PMID: 8460565.


14. Abumi K, Panjabi MM, Kramer KM, Duranceau J, Oxland T, Crisco JJ. Biomechanical evaluation of lumbar spinal stability after graded facetectomies. Spine (Phila Pa 1976) 1990 15:1142–1147. PMID: 2267608.


15. Cooper RG, Mitchell WS, Illingworth KJ, Forbes WS, Gillespie JE, Jayson MI. The role of epidural fibrosis and defective fibrinolysis in the persistence of postlaminectomy back pain. Spine (Phila Pa 1976) 1991 16:1044–1048. PMID: 1835160.


16. Waddell G, Reilly S, Torsney B, et al. Assessment of the outcome of low back surgery. J Bone Joint Surg Br 1988 70:723–727. PMID: 2973465.


17. Sanderson PL, Getty CJ. Long-term results of partial undercutting facetectomy for lumbar lateral recess stenosis. Spine (Phila Pa 1976) 1996 21:1352–1356. PMID: 8725928.


18. Ruetten S, Komp M, Merk H, Godolias G. Full-endoscopic anterior decompression versus conventional anterior decompression and fusion in cervical disc herniations. Int Orthop 2009 33:1677–1682. PMID: 19015851.


19. Ruetten S, Komp M, Merk H, Godolias G. Recurrent lumbar disc herniation after conventional discectomy: a prospective, randomized study comparing full-endoscopic interlaminar and transforaminal versus microsurgical revision. J Spinal Disord Tech 2009 22:122–129. PMID: 19342934.


20. Ruetten S, Komp M, Merk H, Godolias G. Surgical treatment for lumbar lateral recess stenosis with the full-endoscopic interlaminar approach versus conventional microsurgical technique: a prospective, randomized, controlled study. J Neurosurg Spine 2009 10:476–485. PMID: 19442011.


21. Guiot BH, Khoo LT, Fessler RG. A minimally invasive technique for decompression of the lumbar spine. Spine (Phila Pa 1976) 2002 27:432–438. PMID: 11840112.


22. Grob D, Humke T, Dvorak J. Degenerative lumbar spinal stenosis: decompression with and without arthrodesis. J Bone Joint Surg Am 1995 77:1036–1041. PMID: 7608225.


23. Jonsson B, Annertz M, Sjoberg C, Stromqvist B. A prospective and consecutive study of surgically treated lumbar spinal stenosis. Part I: Clinical features related to radiographic findings. Spine (Phila Pa 1976) 1997 22:2932–2937. PMID: 9431629.


24. Katz JN, Lipson SJ, Chang LC, Levine SA, Fossel AH, Liang MH. Seven- to 10-year outcome of decompressive surgery for degenerative lumbar spinal stenosis. Spine (Phila Pa 1976) 1996 21:92–98. PMID: 9122770.


25. Ikuta K, Arima J, Tanaka T, et al. Short-term results of microendoscopic posterior decompression for lumbar spinal stenosis. Technical note. J Neurosurg Spine 2005 2:624–633. PMID: 15945442.


26. Khoo LT, Fessler RG. Microendoscopic decompressive laminotomy for the treatment of lumbar stenosis. Neurosurgery 2002 51(5 Suppl): S146–S154. PMID: 12234442.



27. Haher TR, O'Brien M, Dryer JW, Nucci R, Zipnick R, Leone DJ. The role of the lumbar facet joints in spinal stability: identification of alternative paths of loading. Spine (Phila Pa 1976) 1994 19:2667–2670. PMID: 7899961.


Fig. 1
Endoscope portal and working portal were inserted through the two separated skin incision and docked onto the lamina.
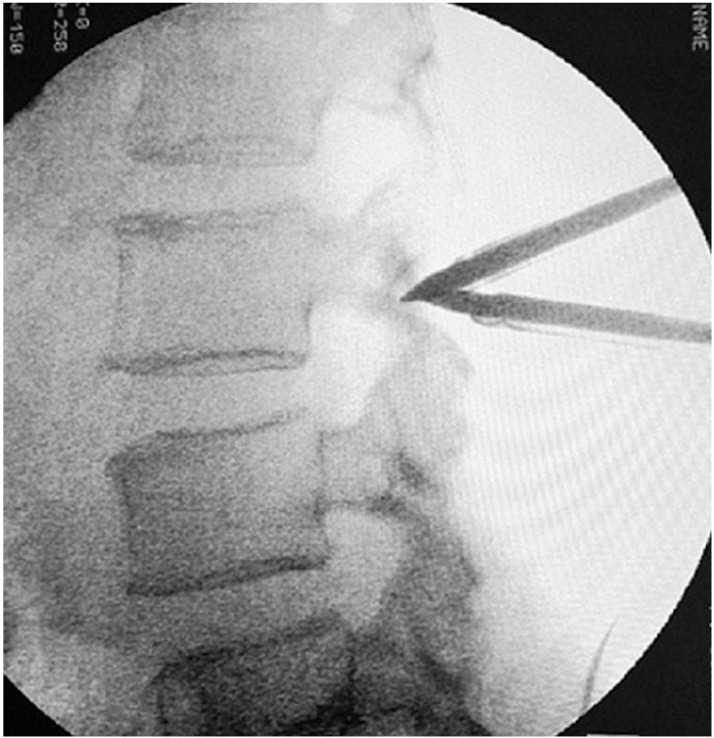
Fig. 2
Potential space was created with endoscopic cautery and evaluated with opaque media fill in urinary catheter.

Fig. 3
Details of the technique. (A) Lamina and ligamentum flavum were seen from potential space. (B) Ipsilateral lamina was remove with high speed burr. (C) Ligamentum flavum of the ipsilateral site was removed. (D) Ipsilateral decompression of nerve root is performed.

Fig. 4
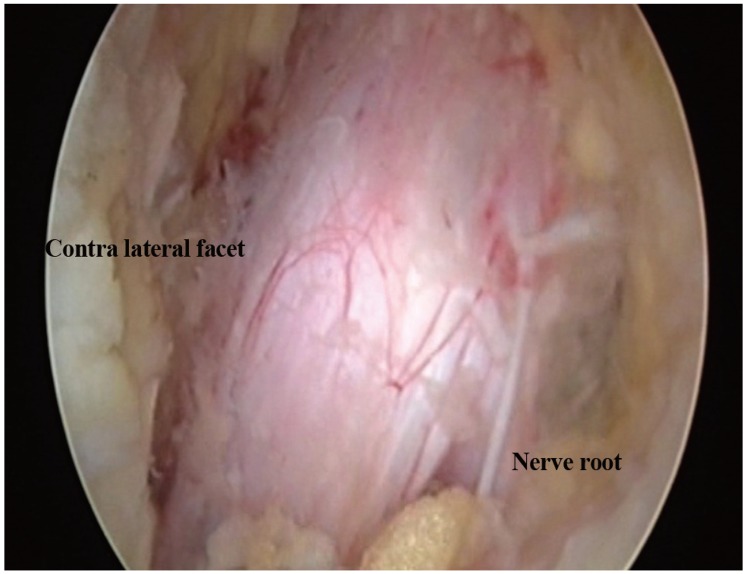
(A) Ipsilateral nerve root mobility was checked. (B) Contralateral ligamentum flavum was excised. (C) Contralateral lamina was removed with rongeur. (D) Contralateral nerve root mobility was checked.

Fig. 6
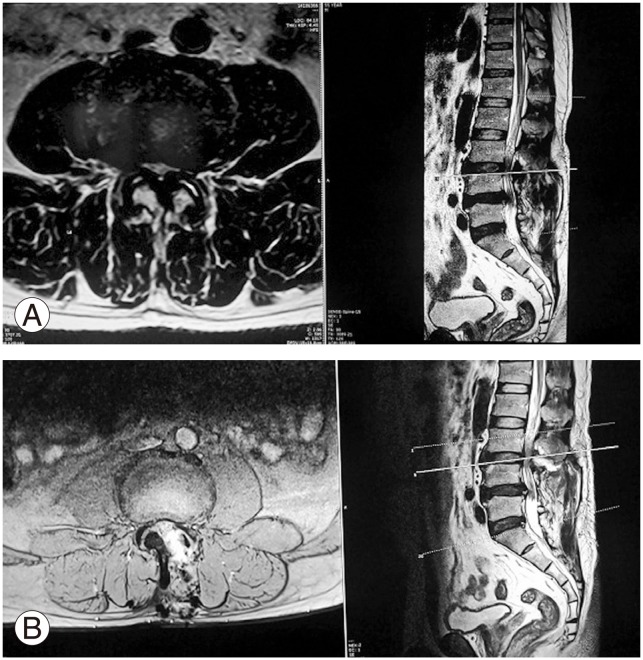
(A) Preoperative magnetic resonance imaging (MRI) of the patient show dural sac compression at L3–4 level. (B) Four weeks postoperative MRI of the same patient show expansion of dural sac.