Rapid Bodyweight Reduction before Lumbar Fusion Surgery Increased Postoperative Complications
Article information
Abstract
Study Design
Retrospective cohort study.
Purpose
To determine the effects of massive weight loss on perioperative complications after lumbar fusion surgery (LFS).
Overview of Literature
Patients who are obese are more likely to experience low back pain, which would require LFS. Nonetheless, they have a higher risk of perioperative complication development compared with individuals who are not obese.
Methods
Patients who underwent LFS at hospitals that participated in the National Surgical Quality Improvement Program database within the United States between 2005 and 2015. Outcomes included 30-day medical complications, surgical complications, and length of stay (LOS). We analyzed a total of 39,742 patients with the use of the International Classification of Disease, ninth revision codes. The patients were categorized in the following two groups: group 1, individuals with a history of massive weight loss within 6 months before LFS, and group 2, individuals without a history of massive weight loss before surgery. Massive weight loss was defined as loss of 10% of total body weight. Patients with a history of malignancy or chronic disease were excluded from the study. Patients in each group were randomly matched based on age, gender, sex, smoking status, and body mass index. Paired two-tailed Student t-tests were used to compare the outcomes.
Results
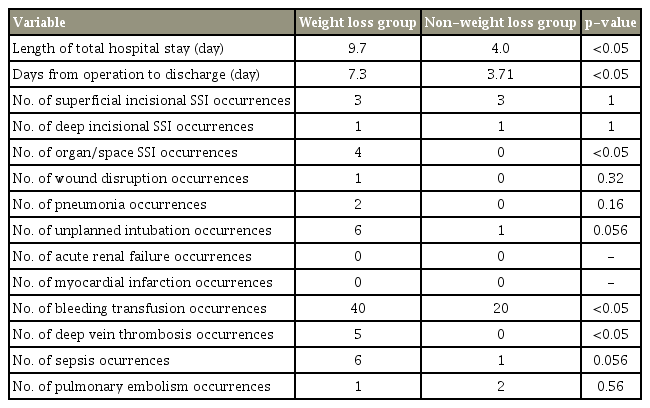
Of the 39,742 patients identified, 129 (0.32%) met the criteria for inclusion in the weight loss group (WL group) and were successfully matched to individuals in the non-weight loss group (non-WL group). Compared with the non-WL group, the WL group had a significantly longer LOS (9.7 vs. 4.0 days, p<0.05), higher surgical site infections (SSIs) (8.0 vs. 3.0, p<0.05), increased number of blood transfusions (40.0 vs. 20.0, p<0.05), and greater deep vein thrombosis (DVTs) (5.0 and 0.00, p<0.05).
Conclusions
On a nationwide scale, rapid weight loss before LFS is associated with a higher rate of postoperative complications, including SSI and DVTs, longer average LOS, and more frequent blood transfusions.
Introduction
Obesity imposes a tremendous burden on the US healthcare system and is a worldwide and national healthcare crisis. On the basis of the Centers for Disease Control and Prevention, obesity prevalence has doubled from 1980 to 2012 [1]. From the spine surgeons’ perspective, this problem is particularly worrisome because obesity is strongly associated with intervertebral disc degeneration and low back pain. As of 2015, obesity’s national prevalence was 26%–32% and low back pain prevalence was between 25.7% and 28.2% [2,3]. With the increased prevalence of obesity and low back pain, the frequency of lumbar fusion surgery also rose. The incidence of spine surgery for low back pain treatment has increased 2.4-fold between 1998 and 2008 with the costs of these surgeries also increased to 7.9-fold [4].
Bariatric surgery, indicated for patients with body mass index (BMI) >40 kg/m2 or >35 kg/m2 with 1 or more significant obesity-related comorbidities when less invasive management has failed, is an increasingly popular treatment for morbid obesity [5]. In 2016, an estimated 216,000 bariatric surgeries were performed, up from 158,000 in 2011 [5]. Bariatric surgery results in remarkable weight loss and helps prevent, improve, and resolve co-morbid conditions, including type 2 diabetes, heart disease, obstructive sleep apnea, and types of cancer. These results are often long-lasting. Patients who underwent bariatric surgery lose an average of 60% of excess weight within 6 months after the surgery and maintain at least 50% of that weight loss 5 years after the surgery [5]. Furthermore, bariatric surgery risks are relatively low because the procedures can be performed laparoscopically. One large report by the Cleveland Clinic in 2006 demonstrated that the complication rate was as low as 0.13% [6].
Patients with obesity, clinically defined as BMI between 25 kg/m2 and 39 kg/m2, are poor surgical candidates because they have increased baseline risk of postoperative complication development, particularly surgical site infections (SSIs) and venous thromboembolism. These complications occur even more frequently in patients with clinically defined morbid obesity, BMI >40 kg/m2. One study conducted an analysis of 84,607 admissions for spinal fusion surgery over a 4-year period and found that although only 1.72% of patients were morbidly obese, they accounted for 97% of in-hospital complications, exhibited longer length of stay (LOS), incurred higher average hospital costs, and had higher mortality rates than non-morbidly obese patients [5].
Given these benefits of bariatric surgery, it is common for the spine surgeons to request that morbidly obese patients who consider elective lumbar fusion surgery for low back pain as bariatric surgery to lose weight before the lumbar surgery. The hope is that weight loss will reduce not only the alarmingly high risk of postoperative complications after spine surgery but also the patient’s back pain, which would alleviate the need for costly surgical intervention that has potential risks and complications. Weight loss alone leads to clinically significant improvement in back pain, although these improvements are often modest and rarely alleviate the need for spine surgery [3,7]. Therefore, patients who underwent remarkable weight loss through bariatric surgery often proceed with lumbar spinal fusion surgery for low back pain; however, they are clinically and metabolically different from other patients who seek lumbar fusion surgery.
Massive weight loss imposes a remarkable metabolic challenge, particularly regarding nutritional deficiencies and the musculoskeletal system. Given this fact, it is important to understand how the increased number of patients who underwent remarkable weight loss before lumbar fusion surgery fare in the perioperative period. This study aims to characterize the postoperative time course of the patients who have recently underwent remarkable weight loss and lumbar spinal fusion for the treatment of low back pain.
Materials and Methods
Our study used the American College of Surgeon’s National Surgical Quality Improvement Program (ACS-NSQIP) database between 2005 and 2015 to identify all the patients who underwent lumbar fusion surgery (CPT codes 22533, 22534, 22558, 22612, 22630, 22633, 22360, 22361, 22370, 22625, 22450, 22830, and 22355, based on the International Classification of Disease, ninth revision). The study did not require the Institutional Review Board approval. All data was obtained through an anonymous registry and informed consent was deemed not necessary by the Institutional Review Board. The ACS-NSQIP is a multicenter prospective registry designed to collect data for improvement purposes. The included hospitals represent academic and community settings in various regions in North America. Over 275 variables were collected, including demographics, preoperative risk factors, procedural indications and details, and 30-day postoperative morbidity and mortality. Data were collected by trained data abstractors and audited for accuracy. The method of ACS-NSQIP abstractor training, data collection process, and reliability audits has been previously reported [8]. Patients who underwent lumbar fusion surgery between 2005 and 2015 were stratified into groups based on whether they had experienced remarkable weight loss. Remarkable weight loss was defined by the ACS-NSQIP use guide as >10% loss of body weight in the 6 months prior to the surgery. Patients with a history of chronic disease or malignancy were excluded. These diseases include diabetes, chronic obstructive pulmonary disease, congestive heart failure, hypertension, acute renal failure, and disseminated cancer. Patients with ascites, those who require dialysis, those with a current wound infection, and those who use steroids were also excluded.
The patients in the weight loss group were matched with the non-weight loss patients. For each patient in the weight loss group, we found patients who had not experience remarkable weight loss and were identical in the following characteristics: age, sex, smoking status, and BMI. Of these identical patients, one was randomly selected and included in the non-weight loss group. Paired two-tailed t-tests were used to compare the surgical outcomes between the weight loss and the non-weight loss lumbar fusion populations. These outcomes included LOS, days from operation to discharge, superficial, deep incisional, and organ/space SSI occurrences, wound disruption, pneumonia, unplanned intubation, acute renal failure, myocardial infarction, bleeding transfusion, deep vein thrombosis (DVT), sepsis, and pulmonary embolism occurrences.
Results
A total of 39,742 patients who underwent lumbar fusion surgery during the study period were identified. Of these, 0.32% or 129 patients met the criteria for inclusion in the weight loss (WL) group. Table 1 presents the difference in postoperative complications and outcomes between the WL group and non-weight loss group (non-WL group). The average post-weight loss BMI in the WL group was 27. Also, the matched patients in the non-WL group had an average BMI of 27 kg/m2.
1. Length of stay
Compared with the non-WL group, the mean LOS and days from operation to discharge were 9.7 versus 4.0 days (p<0.05) and 7.3 versus 3.71 days (p<0.05), respectively.
2. Surgical site infections
The WL group demonstrated a statistically significant trend for organ/space SSI occurrences (4 occurrences versus 0, p<0.05). There was no statistically significant difference between the groups for superficial and deep incisional SSI occurrences (3 versus 3, p=1; 1 versus 1, p=1, respectively).
3. Blood transfusions and deep vein thrombosis
The WL group demonstrated a statistically significant trend for both blood transfusions and DVT occurrences (40 versus 20, p<0.05; 5 versus 0, p<0.05). Chi-square analysis was performed to identify whether the risk of developing wound infections was independent of blood transfusions. Furthermore, there was no statistically significant association between wound infection and blood transfusions. The p-values for this analysis were 0.43 for the superficial infections and 0.64 for the deep wound infections.
4. Other complications
The WL group had more sepsis occurrences, unplanned intubation occurrences, and pneumonia occurrences than the non-weight loss group; however, there were not statistically significant (6 versus 1, p=0.056; 6 versus 1, p=0.056; 2 versus 0, p=0.16). There were two pulmonary embolism occurrences in the non-WL group and one in the WL group (p=0.56). Furthermore, there were no occurrences of acute renal failure or myocardial infarction in either of the group.
Discussion
This study demonstrated a statistically significant association between post-rapid weight loss lumbar fusion surgery and several clinically important postoperative complications. Specifically, the patients who had rapid weight loss prior to spinal surgery had significant organ/space SSI (p<0.05), blood transfusions (p<0.05), DVTs (p<0.05), and longer hospital stays (p<0.05). Although there was a statistically significant difference in organ/space SSI, we did not see a difference in deep SSI. We believe that no statistical difference was found in the deep SSI group because we did not have the power to detect this difference. Only two deep SSIs occurred in both groups combined. Given the patient’s predisposition to infection and limited healing potential, which will be discussed in detail, and the fact that a statistical difference in organ/space SSI was seen, we suspect that a larger study, with more occurrences of deep SSI, would have revealed this difference. Nevertheless, in the context of spine surgery, organ SSI essentially relates to meningitis. Given the NSQIP parameters, in which we used to find the patients that met the criteria, we were unable to differentiate meningitis from other SSIs. This is one of the limitations of this study.
The findings in this study disagree with the results of a recent study by Jain et al. [9], who used a different national surgical database to characterize the risk of perioperative complication development after lumbar spine fusion surgery in patients who previously underwent bariatric surgery. The authors compared the patients with a history of bariatric surgery and diagnosis of obesity at the time of spine surgery with those without a history of bariatric surgery who had a diagnosis of morbid obesity at the time of spine surgery. They reported that the patients in the bariatric group had significantly lower rates of medical complications and SSIs than those in the obese, non-bariatric group [9]. Though the authors of this study were able to precisely identify the patients with a history of bariatric surgery on the basis of the variables collected in the database that was used, the temporal relationship between bariatric surgery and lumbar fusion surgery was unclear. In contrast, although this study utilized the presence of massive weight loss in the absence of malignancy and chronic disease as a surrogate for a history of bariatric surgery, the temporal relationship between weight loss and subsequent lumbar fusion surgery was precisely defined as being within 6 months.
Because the outcomes of spinal surgery after massive weight loss have not been well-studied, it is difficult to propose a time frame in which spinal surgery should be postponed after massive weight loss. Nonetheless, the difference in our results compared with those of Jain et al. [9]’s study may suggest that there is an amount of time after bariatric surgery in which the postoperative complication rate is the same or lower in the patients who have bariatric surgery. Although limited, the data that address the temporal relationship between bariatric surgery and subsequent major surgery demonstrated poorer outcomes in patients who underwent major surgery within 6 months of bariatric surgery. Schwarzkopf et al. [10] reviewed the California State Inpatient Database for all patients who underwent bariatric surgery followed by total joint arthroplasty (TJA) and noted a significant increase in readmission within 90 days in patients who had arthroplasty performed within 6 months from bariatric surgery [10]. Given this information, we suggest that spinal surgery should be scheduled at least 6 months after massive weight loss. Further investigation is needed to determine the exact time period in which it is acceptable to have spinal surgery after massive weight loss.
The perioperative outcomes of obese spine surgery patients who previously underwent bariatric surgery have not been well-studied. Nonetheless, several retrospective trials that examined the relationship between bariatric surgery and TJA have been reported and demonstrate conflicting results. Early studies that investigated this question found a significant reduction in postoperative complications in TJA patients who previously underwent bariatric surgery. Werner et al. [11] reported on 219 morbidly obese patients who underwent bariatric surgery compared with 11,294 morbidly obese controls and demonstrated a major complication rate of half of the control group in the bariatric group (p=0.001) with 40% fewer minor complications (p=0.01). Kulkarni et al. [12] demonstrated similar improvements in post-bariatric TJA patients. Nonetheless, recent studies have been less encouraging and have demonstrated equivalent or higher rates of complications in patients who underwent TJA and who previously underwent bariatric surgery. For example, Severson et al. [13] found no difference in the 90-day complication rates between patients who underwent TJA less than 2 years after bariatric surgery and those who did not undergo bariatric surgery. Similarly, Inacio et al. [14] found that bariatric surgery prior to TJA did not reduce complications or revision rate after TJA. Importantly, this study reported higher rates of readmission (7.2% versus 5.9%) in the bariatric group [14]. Moreover, Martin et al. [15] found that obese patients who previously underwent bariatric surgery had a higher risk of complication after TJA.
Given the high frequency of plastic surgery procedures after bariatric surgery and massive weight loss, several large retrospective and prospective studies of these patients’ postoperative courses have been reported in the plastic surgery literature. One meta-analysis from 2014 analyzed seven retrospective studies that compared the rates of postoperative outcomes between massive weight loss/post-bariatric and non-weight loss/non-bariatric patients who underwent various body-contouring procedures to address the excess skin folds. The authors found that the fixed-effects pooled risk ratio of complication development after body-contouring surgery when comparing post-bariatric and non-bariatric patients was 1.60 (95% confidence interval [CI], 1.30–1.96; p<0.00001; I2=48%) [16]. The findings of an increased risk of postoperative complications in the bariatric group shown in this meta-analysis have been borne out in other large trials that have followed [17,18].
These results are an important reminder that post-bariatric patients typically have metabolic consequences that cannot be ignored, and these surgeries may not improve the surgical risk profile. Deficiencies in various macroand micronutrients that result from bariatric surgery are thought to play an important role in the higher rates of postoperative complications because they interfere with wound healing and the immune system.
A particularly important nutritional deficiency seen after bariatric surgery is hypoproteinemia. Previous reports indicate that up to 25% of bariatric patients are at risk of protein deficiency development after the surgery [16]. Protein deficiency is thought to delay wound healing through impairment of fibroblast proliferation, angiogenesis, and collagen production [19]. Several studies have demonstrated reduced T cell function, decreased phagocyte activity, and lower complement levels in the setting of protein deficiency after gastroplasty [20,21]. One interventional study demonstrated that post-bariatric patients who underwent abdominoplasty and received proteinbased nutritional supplements before body-contouring surgery versus those who did not had a statistically significant lower rate of wound complications after the surgery (n=21) (0% versus 21.8% complication rate, p=0.04) [22]. Nonetheless, there are only a few data in the spine literature that address the effects of similar nutritional supplementation in post-bariatric patients who underwent further surgery.
Additional studies have reported the prevalence of nutrient deficiency in post-bariatric patients to be as high as 30%–50% and 36% regarding iron and zinc deficiency, respectively, even when daily multivitamins are administered [16,23]. Iron deficiency impairs tissue healing and may contribute to fatigue, which increases the risk of DVTs [16]. Finally, severe iron and zinc deficiencies impair collagen production [24] and increase the risk of opportunistic infections [25].
Additionally, fat malabsorption in post-bariatric patients can cause fat-soluble vitamin deficiencies. A total of 69% to 80% of post-bariatric patients present with vitamin A deficiency postoperatively despite daily multivitamin use [26]. Vitamin A is a potent immunostimulant that promotes wound healing through inflammation [16]. Vitamin D and calcium deficiencies are also common after bariatric surgery, with prevalence of vitamin D deficiency ranging from 17% to 63% 18 months to 4 years after the surgery [27]. These deficiencies are clinically relevant and may result in osteomalacia, osteoporosis, and secondary hyperparathyroidism [28]. This is particularly important because hypocalcemia and vitamin D deficiency may impair bone healing after lumbar fusion surgery.
Other common macro- and micronutrient deficiencies found in post-bariatric patients include vitamin B12, B6, and folate, at 3.6%–37%, 17.6%, and 9%–35%, respectively [29]. Vitamin B complexes and folate are essential co-factors in DNA synthesis, and deficiencies in these vitamins may have an effect on cellular proliferation and wound healing. Similarly, megaloblastic anemia from vitamin B12 deficiency may impair wound healing owing to poor oxygen delivery to the healing tissues. Finally, folate and vitamin B12 deficiencies can result in hyperhomocysteinuria—a prothrombotic condition that can result in the development of DVTs. Several studies have demonstrated that elevated homocysteine levels may contribute to DVT development after bariatric surgery [29].
In summary, the findings of this study can be partially explained by the growing body of evidence that suggests a high prevalence of nutritional deficiencies that impact wound healing and immune function after bariatric surgery. These surgeries intentionally interfere with nutritional absorption via reduced nutritional intake and/or impaired absorptive processes along the gastrointestinal tract, and specific macro- and micronutrient deficiencies are therefore common in these patients in the months to years after the surgery. Therefore, compared with patients with adequate nutrient levels, the post-bariatric patients have an increased risk of postoperative complications after surgery subsequent to bariatric surgery.
The limitations of our study include the fact that the NSQIP database only collects data from hospitals in the United States, which limits the international generalizability of our findings. Moreover, the NSQIP database itself is limited in that the data only tracks 30-day morbidity and mortality after a surgery, so patients who developed complications more than 30 days after the surgery would have been missed in our analysis. In addition, the cost of inputting data into the NSQIP from an institutional level is high, so the dataset may disproportionally be constituted of patients who received care at large teaching hospitals. In addition, as with all national datasets, the large heterogeneous population represents patients with different risk factors and the fine detail about these patients is lost in our analysis. Finally, we chose to examine patients who underwent massive weight loss before lumbar fusion surgery as opposed to bariatric surgery specifically. This strategy was chosen because of the manner in which data from the NSQIP database are reported. That is, there is no variable in the NSQIP database to identify weight loss secondary to bariatric surgery. Nevertheless, we do believe that massive weight loss is an appropriate surrogate for post-bariatric surgery. As had been previously stated, patients who underwent bariatric surgery lose an average of 60% of excess bodyweight within 6-month period [5]. Massive weight loss was defined as the loss of greater than 10% of body weight, so patients who underwent bariatric surgery would be captured in our study group. The patients who may be included under “massive weight loss” are those who underwent bariatric surgery, those who had weight loss through natural means (i.e., low-calorie diet and exercise), and those who had weight loss owing to a medical condition. The patients who had massive weight loss because of a medical condition, such as malignancy and chronic diseases, were excluded because we believe that this would eliminate patients in our cohort who have weight loss owing to a medical condition. Additionally, we recognize that it is possible that some patients could be diagnosed with a medical condition that caused weight loss after the 6-month period. Nevertheless, we think that this is unlikely. Most patients who underwent spinal surgery are required to be evaluated by their primary care physician or another general medical doctor for medical clearance before the surgery. It is likely that a medical condition that caused remarkable weight loss would be identified and diagnosed at this time, if it was not previously diagnosed. Moreover, morbidly obese people who lose weight by natural means lose an average of 8% of their body weight in 3–12 months [30]. Thus, on average, these patients do not achieve massive weight loss and would not be included in our study. Therefore, we believe that the majority of the 127 WL patients in our study had weight loss because of bariatric surgery.
Conclusions
On a nationwide scale, weight loss of greater than 10% of body weight within 6 months before lumbar spine fusion surgery is associated with worse postoperative outcomes, including higher organ/space SSI, DVT, increased blood transfusion occurrences, and longer LOS. This difference may be because of the high prevalence of nutrient deficiencies associated with wound healing and immune function seen in post-bariatric patients. Additional studies should therefore seek to further elucidate the reason for these nutritional discrepancies between the experimental and control groups and to compare the efficacy of bariatric surgery alone versus lumbar fusion surgery alone in conjunction with bariatric surgery for the treatment of low back pain.
Notes
No potential conflict of interest relevant to this article was reported.