Effectiveness of Neurodynamic Mobilization versus Conventional Therapy on Spasticity Reduction and Upper Limb Function in Tetraplegic Patients
Article information
Abstract
Study Design
The study employed a pre- and post-test experimental design.
Purpose
This study was designed to assess the effect of neurodynamic mobilization of the median nerve on upper limb spasticity in tetraplegic patients.
Overview of Literature
Spasticity is a common and potentially disabling and bothersome complication in patients with spinal cord lesion; this disorder can negatively influence the quality of life by restricting the patient’s ability to perform activities of daily living. Neural mobilization is currently used for reducing the spasticity in individuals with neurological disorders.
Methods
Twenty subjects with traumatic spinal cord injury (level C5–C8) and upper limb spasticity in the finger and wrist flexors were enrolled. They were randomly allocated to two different groups using a computer-generated randomization schedule: group I comprised the neurodynamic mobilization group (n=11) and group II was the conventional therapy group (n=9); the subjects were administered therapy for 5 days every week for a period of 4 weeks. Upper limb spasticity was assessed using the Modified Ashworth Scale for wrist and finger flexors; F-wave amplitude, latency, and F-wave/M-wave amplitude ratio (F/M ratio) were examined using the F-wave scores of the median nerve; and upper limb function was determined using the Capabilities of Upper Extremity (CUE) Questionnaire.
Results
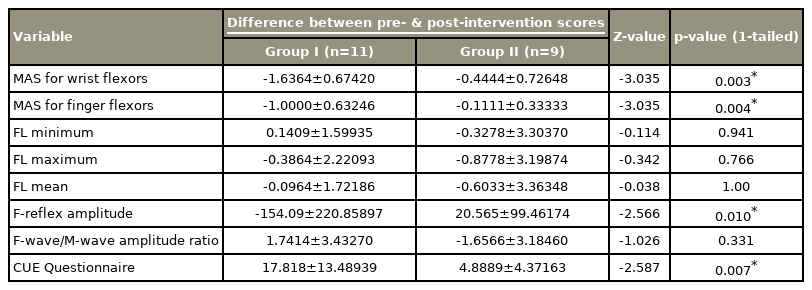
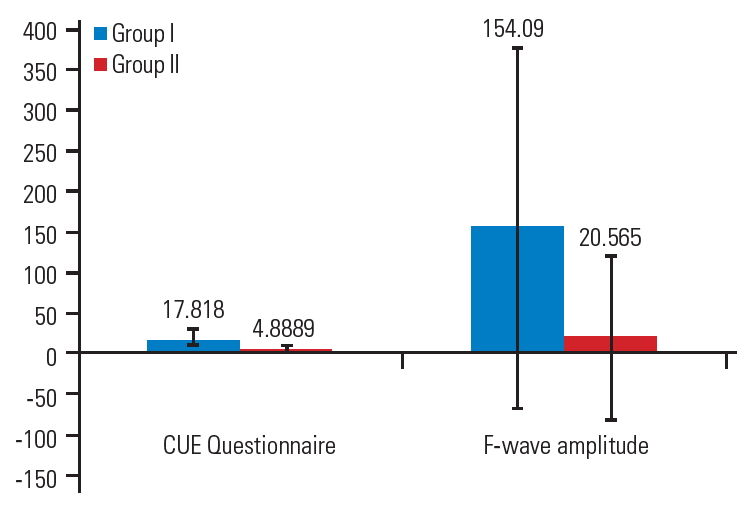
After 4 weeks of intervention, between-group comparisons showed a significant difference in the pre-intervention and postintervention scores on the Modified Ashworth Scale score for wrist flexors (−1.64±0.67), Modified Ashworth Scale score for finger flexors (−1.00±0.63), F-wave amplitude (−154.09±220.86), F/M ratio (−0.18±0.24), and CUE scores (17.82±13.49).
Conclusions
These results suggest that neurodynamic mobilization of the median nerve may be effective for upper limb spasticity control and upper limb functional improvement in tetraplegic patients.
Introduction
Spinal cord injuries (SCIs) interrupt the axonal pathways and segmental spinal cord connections at and below the level of the injury, leading to severe motor, sensory, and autonomic dysfunction [1]. SCI patients experience many major complications, depending on the level and severity of the lesion, such as paralysis, spasticity, sensory loss, intractable pain, pressure sores, and urinary and other infections that lower their health-related quality of life [2]. Spasticity is a common complication followed by SCI that potentially limits functional independence [3].
Spasticity is reported to be one of the most difficult health complications after SCI [4]. Moreover, 65%–78% of subjects with chronic SCI (≥1 year post injury) show symptoms of spasticity [5]. The most common anatomical region of the injury was the cervical spine (43.9%–61.5%) causing tetraplegia [5]. Moreover, 60% of those with a cervical injury had spasticity in their upper extremities. Spasticity can negatively affect the quality of life by restricting activities of daily living (ADLs); inhibiting efficient walking and self-care; causing pain and fatigue; disturbing sleep; and hampering safety, leading to the development of contractures, pressure ulcers, infections, and negative self-image, thus complicating the role of the caretaker and impeding rehabilitation efforts [5]. The major concern in cervical SCI patients is upper limb spasticity because it can cause barriers in restoring function by hampering the ADLs that, in turn, adversely affect the level of independence [6]. The management of spasticity is desirable for the control of passive problems, like reducing pain, facilitating splint wearing, easing positioning and hygiene, and preventing contractures, or functional problems, including the individual’s decreased ability to perform important motor functions [5].
Systemic pharmacological treatment of spasticity is frequently prescribed for patients with SCI, including baclofen, tizanidine, and gabapentin; these may have possible adverse effects, such as dizziness, nausea, withdrawal seizures, hallucinations, and ataxia; no single medication has shown a positive effect in all patients [4]. Rehabilitation is considered necessary in the management of spasticity as a long-term regimen along with surgical and pharmacological interventions. The goal is to diminish spasticity and allow voluntary movements and/or to improve the ability to independently perform ADLs, such as transfers, dressing, and toileting [4].
Neural mobilization is a currently used technique that aims to reduce the spasticity in patients with neurological disorders. Neural mobilization refers to a group of techniques that aim to place the neuraxis in tension and stretch it with appropriate mobilization through certain postures, along with the application of slow, rhythmic movements of the joints intended to reach the peripheral nerves and the spinal cord [7]. These techniques are developed gradually, using the diagnostic tests proposed by Elvey and Hall [8] to assess adverse neural tension.
The main purpose of neural mobilization is to restore the dynamic balance between the movement of neural tissue and mechanical connections of the surroundings, to promote optimal physiological function, and to restore the normal mechanical and physiological state of motion and posture [7,9]. These movements allow the maintenance, elasticity, and extensibility of the nervous system, thus enhancing muscle maintenance and extensibility [10].
F-wave is a compound action potential elicited by the supra-maximal antidromic stimulation of a motor nerve after the direct muscle response. They are useful in the assessment of proximal conduction slowing, as evident in spasticity [11]. Rosche et al. [12] showed that F-wave amplitude and F-wave/M-wave amplitude ratio (F/M ratio) can be used to document spasticity.
This study was designed to test the hypothesis that neurodynamic mobilization is effective in reducing spasticity of the upper limbs and improving hand function in tetraplegic patients.
Materials and Methods
The study employed a pre- and post-test experimental design.
1. Participants
We used convenience sampling to enroll 22 tetraplegic subjects with traumatic SCI in our study as per the inclusion and exclusion criteria. The subjects were recruited from the rehabilitation department, Indian Spinal Injuries Centre, Vasant Kunj, New Delhi after they provided written consent for study participation. Twenty subjects completed the intervention successfully, and two subjects dropped out (one dropped out after 2 days of treatment and the other after 4 days due to their inability to complete the study). One hand of each subject was given the intervention based on the inclusion/exclusion criteria. In patients for whom both hands met the criteria, the hand with greater spasticity was chosen for the study.
2. Inclusion criteria
Subjects with traumatic SCI [13]; with American Spinal Injury Association (ASIA) impairment grade A, B, C, and D [4]; those who could lie in the supine position [2]; those who were oriented and alert; those aged 18–65 years [6]; those with non-progressive SCI and residual neurological deficits [14]; and those with complete or incomplete SCI were enrolled.
3. Exclusion criteria
Subjects with a score >3 on the Modified Ashworth Scale [2]; those who did not provide signed informed consent [2]; those with symptomatic zygapophyseal joints of the cervical spine [15]; those experiencing dizziness [15]; those with pathologies that affected the nervous system, such as diabetes, multiple sclerosis, and Guillian–Barre syndrome; those with a recent history of any surgery [15]; and subjects with Cauda Equina lesions [15] were excluded.
4. Procedure
A detailed explanation of the study and a detailed patient information sheet was given to all the subjects. Written informed consent was obtained from all the study subjects. The study protocol was reviewed and approved by the Institutional Ethical Committee (ISIC/IIRS/RP/2015/068).
Demographic data, neurological details, and baseline characteristics of the subjects were collected using the evaluation proforma, ASIA form for SCI to determine the neurological level of the SCI and International Spinal Cord Injury Core Data Set form. F-wave for stimulating median nerve (abductor pollicis brevis muscle) was performed by an experienced neuro-electrophysiological technician working at the neuro-electrophysiological department of Indian Spinal Injuries Centre. Subjects were then randomly and equally divided into the following groups using a fixed random allocation method: group Ι comprised the neural mobilization group and group ΙΙ was the conventional group. Before the intervention, subjects from both the groups were assessed by a blinded assessor, and scores were obtained for the following outcome measures: (1) scores for spasticity of the upper limb of choice (Modified Ashworth Scale score for wrist flexors and finger flexors); (2) score on Capabilities of Upper Extremity (CUE) Questionnaire; and (3) F-wave parameters (F-wave amplitude; minimum, maximum, and mean latency and; F/M ratio).
Group Ι underwent neurodynamic mobilization of the median nerve. Mobilization was performed for all the subjects for 12 minutes during each session; sessions were conducted 5 times each week for 4 weeks (total 20 sessions) from the time of study initiation. Each subject was made to lie in the supine on the plinth with the scapula free of the bed. With the maintenance of shoulder girdle depression, the glenohumeral joint was extended, abducted, and laterally rotated; the elbow was extended; the forearm was supinated; and the wrist, fingers, and thumb were extended. After holding this position, neural mobilization was performed with slow, rhythmic oscillations of wrist flexion and extension. Twenty oscillations were performed each minute for 3 minutes; the process was performed thrice during the same session, with a 1-minute interval between consecutive attempts [13].
Group ΙΙ received conventional therapy. Stretching was performed for all subjects for 15 minutes in every session; we conducted sessions 5 times every week for a period of 4 weeks (total 20 sessions) from the time of study initiation. Each subject was made to lie in the supine position on the plinth with the scapula free of the bed. The subject’s shoulder was in abduction, elbow in extension, wrist in dorsiflexion, and fingers in extension. After holding this position, stretch was maintained for 1 minute and repeated 9 times during every session [13]. The assessor was blinded to the type of intervention given to the subjects. All the interventions were provided by the same therapist. Post-intervention scores were obtained after 28 days of study initiation, that is, on the 29th day of the study for all the outcome measures in the same manner as that during the pre-intervention assessment.
All the data were analyzed using IBM SPSS ver. 21.0 (IBM Corp., Armonk, NY, USA). Wilcoxon signedrank test was used for within-group analyses, and Mann-Whitney U-test was used for between-group analyses. Statistical significance was set at p≤0.05, and the value of the confidence interval was set at 95%.
Results
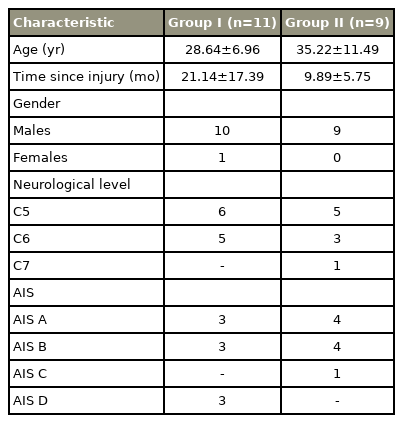
A total of 20 subjects participated in the study. The demographic characteristics of the study population are summarized in (Table 1).
1. Within group analysis
Our results showed significant differences between the pre-intervention and post-intervention scores of the Modified Ashworth Scale for wrist and finger flexors, F-wave amplitude, and scores of the CUE in group I and scores of the CUE scores in group II.
2. Between-group analysis
There were significant differences between the pre-intervention and post-intervention scores of the Modified Ashworth Scale for wrist and finger flexors, F-wave amplitude, and score of the CUE in group I (Table 2, Figs. 1, 2).
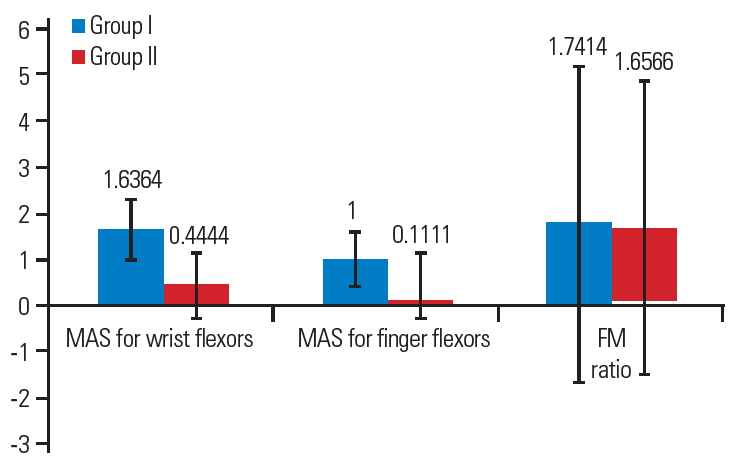
Mean difference of Modified Ashworth Scale score (MAS) for wrist & finger flexors and F-wave/M-wave amplitude (F/M) ratio scores.
Discussion
The present findings were consistent with a previous report by Solorzano [16] as per which neurodynamic mobilization of the median nerve is effective in decreasing the spasticity in upper limb muscles of SCI patients.The neurodynamic mobilization treatment administered in this study was adapted from the study by Solorzano [16] that used the findings of two studies conducted by Godoi et al. [17] and Castilho et al. [7].Castilho et al. [7] found a significant decrease in the electromyography activity of the biceps brachii immediately after the intervention of neural mobilization of the median nerve in stroke patients. Solorzano [16] found a significant reduction in the Modified Ashworth Scale score, improvement in the joint range of motion, Action Research Arm Test and in Functional Independence Measure after 3 weeks of neural mobilization in a case study on a quadriplegia patient. Consistent with the results of both studies, clinically meaningful improvements were observed in the F-wave amplitude, F/M ratio, Modified Ashworth Scale score, and CUE score. Our results were in agreement with those reported by Solorzano [16] and suggested that a longer intervention program may be more effective.
As Butler and Jones [18] stated “central and peripheral nervous systems are considered one system upheld by three dimensions: mechanical, electrical, and chemical continuity”; it is assumed that after an injury to the nervous system, tension increases and negatively affects the patient’s mobility and functional ability. If the neural tension increases unusually, the muscle tone is altered and is markedly increased in the distal segments of the extremities [16].
Marinzeck [19] stated that the use of upper limb neurodynamic test 1 improves retrograde axoplasmic flow, which is abnormal in spasticity, thereby alleviating nerve tension, reducing restrictions and adhesions of the nerve to the surrounding tissues, thus improving the conditions for the enhancement of activities and muscle nutrition. The hypothetical benefits of neurodynamic mobilization include facilitating nerve sliding, reducing neural adhesion, dispersing harmful liquids, increasing nerve vascularization, and improving axoplasmic flow [19]. All these physiological functions of neural tissues are compromised in patients with spasticity; therefore, we can conclude that neurodynamic mobilization reduces spasticity. This is consistent with the results of our study wherein neurodynamic mobilization reduced spasticity, as shown by the reductions in the F-wave amplitude, F/M ratio, and Modified Ashworth Scale scores.
The upper limb function improved in both, the neural mobilization group and the conventional therapy group. However, the upper limb function scores, including the CUE score, showed more significant improvements in the neural mobilization group than in the conventional group. Cowell and Phillips [20] in 2002 reported that the nerve mobilization technique improves the nervous system structure and muscle flexibility [21]. Similarly, we found that improvement in median nerve flexibility might contribute to improved upper limb function. The improvement in the CUE score in our study was similar to that reported by Cha et al. [22] in 2014. In his study on 22 stroke patients, he found that sciatic nerve mobilization improved lower limb function after 4 weeks of sciatic nerve mobilization intervention [22].
The conventional therapy group showed no significant differences in spasticity. Neurodynamic mobilization is an effective method to resolve the issue of spasticity because it is less time consuming unlike stretching and weight bearing, is cost effective, and does not need any equipment or machine.
Although this study showed that neural mobilization helps reduce spasticity, the study also has certain limitations. First, we employed a relatively small sample size. Second, only trauma cases of SCI were included. Therefore, our results cannot be generalized to non-trauma cases. Finally, we did not perform long-term follow up.
Conclusions
Neurodynamic mobilization of the median nerve is more effective than conventional therapy in reducing upper limb spasticity in patients with traumatic SCI, as measured using F-wave and the Modified Ashworth Scale. The upper limb function, as measured using the CUE score, was improved in both the groups.
Notes
No potential conflict of interest relevant to this article was reported.