Anterior Decompression and Fusion for the Treatment of Cervical Myelopathy Caused by Ossification of the Posterior Longitudinal Ligament: A Narrative Review
Article information
Abstract
Occasionally, ossification of the posterior longitudinal ligament (OPLL) causes cord compression, resulting in cervical myelopathy. OPLL differs from other causes of cervical spondylotic myelopathy in several ways, and the surgical strategy should be chosen with OPLL’s characteristics in mind. Although both the anterior and posterior approaches are effective surgical methods for the treatment of OPLL cervical myelopathy, they each have their own set of benefits and drawbacks. Anterior decompression and fusion (ADF) may improve neurological recovery, restore lordosis, and prevent OPLL mass progression. The benefits can be seen in patients with a high canal occupying ratio or kyphotic alignment. We discussed the benefits, limitations, indications, and surgical techniques of ADF for the treatment of OPLL-induced cervical myelopathy in this narrative.
Introduction
Ossification of the posterior longitudinal ligament (OPLL) causes spinal cord compression, resulting in myelopathy [1–3]. Histologically, it is caused by fibroblasts and chondroblasts proliferation as well as intramembranous ossification [4,5]. OPLL risk factors include diabetes, obesity, diffuse idiopathic skeletal hyperostosis, and ankylosing spondylitis [6]. OPLL is frequently found in people from East Asian countries such as Korea and Japan [7,8], with prevalence rates ranging from 1.9% to 4.0%. Its prevalence in North America and Europe ranges from 0.1% to 1.5% [7–9]. OPLL progresses slowly; 42% of OPLL lesions show increasing thickness and 82% show longitudinal progression after 10 years of follow-up [8,10].
Because OPLL most commonly affects the cervical spine, many surgical strategies for treating OPLL-induced cervical myelopathy have been proposed [3,11,12]. Anterior decompression and fusion (ADF) techniques include anterior cervical discectomy and fusion (ACDF) and anterior cervical corpectomy and fusion (ACCF), while posterior operative techniques include laminoplasty and laminectomy/fusion [13–15]. A novel anterior translational osteotomy technique has recently been reported [16,17]. When deciding on an appropriate surgical strategy, many factors must be considered including the shape of the OPLL lesion, number of levels involved, presence of neck pain, and sagittal alignment [18]. Surgical treatment of OPLL-induced cervical myelopathy has been linked to more complications than treatment of cervical spondylotic myelopathy, and failure to select the best surgical method could result in poor clinical outcomes [19].
Previous research has reported the benefits and drawbacks of anterior and posterior surgical strategies for OPLL-induced cervical myelopathy [11,12,20]. Because OPLL shape and sagittal alignment vary so greatly between patients, applying strict algorithms or indications for surgical method selection is difficult [21]. Before surgery, surgical strategies should be tailored to the patient’s specific needs, and the advantages and disadvantages of each method should be thoroughly discussed. We focused on anterior surgical strategies for OPLL-induced cervical myelopathy in this narrative review, discussing their benefits, limitations, indications, and techniques.
Advantages of Anterior Decompression and Fusion
Although laminoplasty achieves cord decompression through indirect decompression and posterior shift of the spinal cord [22], ADF can achieve direct decompression by removing the offending OPLL mass [23]. As a result, ADF is expected to result in better neurological recovery than posterior surgery [3,23]. Chen et al. [23] demonstrated that in OPLL patients involving ≥3 levels or with a canal occupying ratio of ≥40%, ADF significantly improved Japanese Orthopedic Association (JOA) score recovery compared to laminoplasty. Another factor that contributes to better neurologic outcomes is the restriction of cervical motion by ADF. According to Saito et al. [24], large segmental motion at the peak of the OPLL mass is a significant factor influencing poor surgical outcomes, and ADF should be considered when large preoperative segmental range of motion (ROM) is detected.
In terms of sagittal alignment, ADF is superior to posterior surgeries because it can restore cervical lordosis, even in patients with rigid kyphosis [25,26]. When an OPLL mass prevents lordotic alignment with neck extension, it is often necessary to break or remove the ossified mass to restore cervical lordosis [27,28]. Although ADF can directly manipulate the ossified mass, lordosis restoration is only possible with flexible kyphosis, posterior decompression, and fusion [29,30]. Furthermore, postoperative kyphosis due to neck musculature injury has been reported with laminoplasty [31,32]. Although this study did not report C2–C7 lordosis measurements, Iwasaki et al. [25] reported that ADF improved cervical lordosis [33]. Park et al. [34] reported in their retrospective study that ADF restored lordosis better than laminoplasty and that the increased C2–C7 sagittal vertical axis with ADF was less than that obtained with posterior surgery. Since the loss of lordosis and increased positive balance are associated with a negative correlation with health-related outcomes, ADF can provide better results than posterior surgery because it can restore proper sagittal alignment [35,36].
Although ADF is usually used to remove OPLL directly, a complete removal is not always possible. Another advantage of ADF over laminoplasty for treating OPLL cervical myelopathy is that it inhibits OPLL growth even when OPLL lesions are not completely removed [13,37]. Many studies have found that OPLL continues to progress after laminoplasty with an incidence ranging from 38.9% to 73.0% [8,31]. Although clinical deterioration as a result of OPLL progression is uncommon following laminoplasty, delayed neurologic deterioration has been reported. It has been proposed that ≥5° increase in segmental ROM is a risk factor for OPLL progression [38]. Because ADF eliminates segmental motion, the progression of ossified mass is expected to be slower than in non-fusion procedures such as laminoplasty [38,39]. According to Lee et al. [39], radiological OPLL progression was 62.5% with laminoplasty and 7.6% with fusion operations including. According to Lee et al. [37], OPLL thickness progression was significantly less with ADF than with laminoplasty.
Finally, for patients with significant preoperative neck pain, ADF is beneficial over laminoplasty [40–44]. ACDF, according to Stull et al. [42] significantly improves neck pain, regardless of the etiology. According to Massel et al. [43] regardless of presenting symptoms, ACDF can result in significant improvement of neck pain. However, when compared to ADF, posterior operations are invariably associated with more extensive dissection and neck muscle injury as well as aggravation of neck pain [41]. Kimura et al. [40,41] reported a 32% incidence of significant neck pain 2 years after laminoplasty, and proposed that moderate-to-severe baseline neck pain and anterolisthesis are risk factors for postoperative neck pain aggravation [41].
Complications and Limitations
When compared to laminoplasty, ADF has a significantly higher complication rate when treating OPLL [11,13,18,26]. OPLL patients were found to be at a higher risk of perioperative complications than those with other types of degenerative cervical spondylosis [19]. Cerebrospinal fluid (CSF) leakage, dysphagia/dysphonia, hematoma, and hardware-related complications are the most common complications of ADF [45]. Furthermore, although rare, other anterior approach-related complications such as esophageal perforation or recurrent laryngeal nerve palsy can occur with ADF.
Dural tear and CSF leakage are common in ADF surgeries with rates ranging from 6.3% to 31% [46,47]. Such complications, if not treated properly, can result in pseudomeningocele formation, meningitis, or persistent fistula formation [48]. OPLL masses frequently adhere to the dura, and in severe cases, ossification extends into the dura (dura ossification), as indicated by the double-layer sign [49]. This sign, which is characterized by the anterior ligamental and posterior dural rims of hyperdense ossification separated by a central hypodense mass on an axial CT, is considered the most significant risk factor for the development of CSF leak [50]. Yang et al. [49] reported that when dural ossification has a long-straight shape rather than a crescent shape, a dural tear is unavoidable and a posterior approach should be considered. Yamaura et al. [51] proposed the “floating technique” to reduce CSF leaks during anterior decompression, which involves thinning the ossified mass with a high-speed burr and releasing the OPLL lesion from the vertebral bodies, allowing an anterior float of the remaining OPLL lesion. Joseph et al. [52] found that using the floating technique reduced the occurrence of dural tears by 6.3%. Although primary repair of CSF leaks is recommended, ADF may not always be able to perform it due to limited working space [45]. When the primary repair was not possible, several authors proposed a step-by-step approach for dural tears [53,54]. Moon et al. [53] proposed blocking the tear site with an artificial dura and fibrin glue. In their case series of seven patients, two recovered without the use of a lumbar drain or wound-related complications, while the others had pseudomeningocele or wound problems [53]. According to Fengbin et al. [54] combining intraoperative gelatin foam/fibrin glue, postoperative bed rest, adequate hydration, and lumbar drain successfully treated 11 patients with CSF leak after ADF for OPLL cervical myelopathy without revision surgery.
Postoperative dysphagia after ADF ranges from 1.4% to 60.0%, and chronic dysphagia lasting >3 months ranges from 0% to 18.0% [45,55,56]. Female sex, revision surgery, and >2-level operations have all been linked to higher rates of postoperative dysphagia [57]. Several studies found that using local intraoperative steroids reduced the rate of dysphagia following ADF [58]. The incidence of postoperative hematoma after ADF ranges from 0.0% to 1.9% [46]. According to a retrospective study, ADF for OPLL increases the risk of hematoma [59]. When compared to spondylotic myelopathy surgery, hardware complications such as subsidence, screw migration, or plate breakage were not increased with ADF for OPLL [45]. Although such instrument-related complications occasionally necessitate revision surgery, particularly in ACCF, careful observation is usually sufficient in mild cases due to a benign clinical course [60]. C5 palsy is less common with ADF than with posterior surgery [61,62]. Patients with OPLL, on the other hand, have a higher rate of C5 palsy than those with spondylotic myelopathy [62]. According to a meta-analysis, the C5 palsy rate after ADF for OPLL was 5.5% [61].
Surgical Indications
While ADF can achieve optimal results through direct decompression and cervical lordosis restoration, it is technically demanding and has a higher rate of complications [26]. Given the benefits and limitations of ADF, it should be performed only when the expected benefits outweigh the risks of complications such as CSF leakage, graft dislodgement, hematoma, or pseudarthrosis. Laminoplasty is still a viable surgical option that can be used safely on the majority of OPLL patients with cervical myelopathy [63].
K-line (−) cases are one of the indications for ADF in the treatment of OPLL-induced cervical myelopathy [32,64]. A K-line is defined as the line connecting the C2 and C7 spinal canal midpoints [65]. The K-line state is defined as (−) when the OPLL lesion extends posterior to the K-line, and (+) when the peak of the ossified mass remains anterior to the K-line [65]. The K-line state can be affected by both the cervical alignment and canal occupying ratio of the ossified mass, with kyphotic alignment or a high canal occupying ratio leading to a K-line (−) state [66]. Many studies have suggested that performing laminoplasty on K-line (−) patients is associated with poor neurological recovery because posterior spinal cord shift may not occur [65]. According to Koda et al. [67], for K-line (−) patients, ADF achieved higher JOA score recovery (72.5%) than laminoplasty (14.4%) or posterior decompression and fusion (43.6%) [64]. They also discovered that postoperative conversion of the K-line state from (−) to (+) is an independent favorable prognostic factor for neurologic recovery. As a result, if planned ADF surgery can change the K-line state into (+) by removing the ossified foci or restoring cervical lordosis, it can improve outcomes [32,67]. The “Kappa line” has also been suggested, which is defined as a straight line connecting the center of the spinal canal at one level above and below the levels where decompression is required, has been proposed as a modification of the K-line named [66]. It has been proposed as an indicator that could replace the K-line function when OPLL lesions span a small number of levels and have a better correlation with neurologic recovery, though this is a preliminary suggestion that requires further validation [66].
ADF is also indicated in cases where the canal occupying ratio is high [11,25,26]. Iwasaki et al. [25] proposed that ADF has a higher JOA recovery rate, especially when the canal occupying ratio is ≥60%. Kim et al. [3] reported that ADF can provide better neurologic outcomes than laminoplasty in patients with a canal occupying ratio of ≥60% and cord intramedullary increased signal intensity. These studies also suggest that when the canal occupying ratio is <60%, the JOA recovery rate is comparable to ADF or laminoplasty. Hill-shaped OPLL lesions are another indication of ADF [25]. Hill-shaped lesions, according to Iwasaki et al. [25], are a risk factor for poor recovery after laminoplasty. Moon et al. [68] found that when OPLL lesions were hill-shaped rather than plateau-shaped, the JOA recovery rate was significantly higher with ADF than with laminoplasty.
The number of levels involved in the OPLL mass should also be considered. According to Liang et al. [69], an increased number of surgical levels and OPLL are risk factors for increased postoperative bleeding, which may increase the risk of hematoma. Graft extrusion is common in patients undergoing three-level ADF, according to Sasso et al. [70]. Due to the high rate of graft-related complications, they also suggested that posterior fixation should be performed in conjunction with a multi-level ACCF. The ACCF construct has a long lever arm, which may cause graft migration, especially when ≥3 are involved [20]. Given the increased risk of graft-related complications or hematoma, Ha et al. [20] recommended ADF when ≤2 vertebral body levels are involved.
Finally, if a patient has significant preoperative neck pain, ADF may be the best option [40,42,43]. As previously stated, ADF can alleviate neck pain, whereas the posterior approach can aggravate neck pain [40,42,43].
Surgical Techniques
1. Anterior cervical discectomy and fusion
Although ACDF is the most commonly used anterior surgical method, it has significant limitations for OPLL myelopathy cases due to limited working space [71]. OPLL usually involves multiple levels of the vertebral body, and ACDF for focal OPLL lesions limited to the motion segment is performed [71,72]. Furthermore, ACDF for OPLL myelopathy is technically demanding due to the limited operative space may increase the risk of CSF leak and iatrogenic neurologic deterioration [73]. ACDF, on the other hand, causes less intraoperative bleeding, a shorter hospital stay, and better cervical lordosis restoration than ACCF [74]. As a result, several technical variations for improved visualization of the OPLL lesion have been reported to maximize the benefits of ACDF while enabling successful decompression [75].
The “Williams-Isu method” is an alternative technique that can improve the visualization of OPLL lesions in ACDF [76,77]. The inferior part of the cranial vertebra and the superior part of the caudal vertebra are removed with a boxed-shaped osteotomy [76,77], allowing for a larger working space [76,77]. Following decompression, bone blocks extracted from the vertebral bodies are rotated 90° and inserted as intervertebral spacers into the disc space [76,77]. Because the removed box-shaped portion of the vertebra is used as a bone graft, no additional interbody spacers are required [76,77].
Several authors have reported oblique wedge-shaped osteotomies combined with ACDF for improved visualization [75,78]. Lei et al. [75] described an “enlarged ACDF” modification of the ACDF. Following complete discectomy, a wedge-shaped osteotomy is performed with a burr or osteotome to widen the visualization of the OPLL [75]. OPLL lesions are thinned by drilling and removed with a micro cuvette [75]. Zekaj et al. [78] also demonstrated that oblique osteotomy performed after discectomy could increase the working space available for removing the ossified mass. However, these techniques cause endplate damage and have been linked to increased subsidence [75,78]. It is necessary to propose a technique that preserves the anterior portion of the endplate while performing wedge-shaped osteotomies only at the posterior aspect of the disc space (Fig. 1).

Case of oblique posterior endplate cutting for removal of ossification of the posterior longitudinal ligament (OPLL). A 50-year-old male presented with gait disturbance and hand clumsiness. (A) Preoperative sagittal magnetic resonance imaging (MRI) demonstrates significant cord compression with cord signal change at C5–6 level. (B) Preoperative computed tomography (CT) shows that segmental OPLL is the offending structure causing cord compression. (C) Anterior cervical discectomy and fusion, C5–6 was performed. (D) Posterior portion of endplates were drilled obliquely, which enabled the successful removal of the ossified mass extending behind the vertebral body. (E) Successful decompression is confirmed in postoperative MRI. (F) Postoperative 1-year CT demonstrates successful fusion without significant subsidence.
ACDF can also be combined with posterior laminectomy and fusion to restore lordosis [27,28]. Two retrospective studies described a 3-staged operation technique that involves breaking the OPLL mass with ACDF allowing the kyphotic deformity to be flexible [27,28]. Following that, cervical lordosis is restored by inserting multiple lordotic-shaped interbody spacers and rod connections [27,28]. Decompression is then achieved by laminectomy and changing the K-line (−) into (+) state [27,28].
2. Anterior cervical corpectomy and fusion
As an anterior decompression method, ACCF has traditionally been the most widely used surgical strategy for OPLL cervical myelopathy [3,23]. It has a larger working space than ACDF and allows for complete visualization of the OPLL mass [79]. Following partial removal of the vertebral bodies using a rongeur, the posterior cortex of the vertebrae is thinned as much as possible with a burr. A micro-dissector is then used to separate the OPLL from the dura. When OPLL is associated with dura ossification, the anterior floating method is used to carefully preserve the dural portion to avoid dural tears [51]. According to Wang et al. [79], performing a wider decompression while performing discectomies and vertebral body removal can expose the lateral margin of the OPLL mass, allowing for safer OPLL removal (Fig. 2).
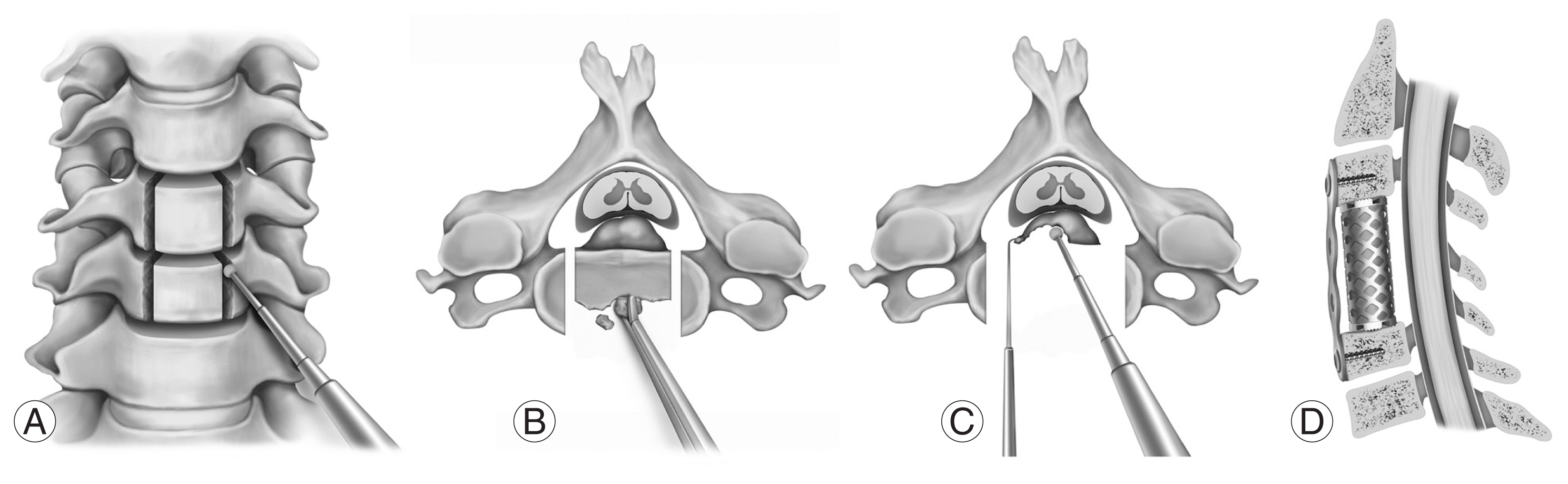
Surgical procedures of an anterior cervical corpectomy and fusion. (A) The lateral margin of the corpectomy should be drilled to ensure a safe corpectomy. The base of the uncinate process after discectomy is the landmark, as a narrower corpectomy could result in an insufficient decompression, whereas a wider corpectomy could cause a vertebral artery injury. (B) The vertebral body is removed until the posterior cortex and ossified mass are visualized. (C) The ossified mass is removed by careful drilling. The careful use of a micro-curette and Kerrison punch at the final stage of removal could prevent a dural tear. When a floating method is planned, the ossified mass does not need to be drilled completely. However, the proximal, distal, and lateral margins of the ossified mass should be sufficiently visualized and ossification of the posterior longitudinal ligament should be mobilized by cutting all the surrounding edges of the ossified mass. (D) Reconstruction is performed using a tricortical graft/cage and anterior cervical plate. Care must be taken to avoid over-distracting the segment, as this may compromise the postoperative stability.
Since ACCF is frequently associated with subsidence or graft dislodgement, the stability of the ACCF construct has been a source of concern [80]. The ACCF construct has a long lever arm and few fixation points, which can cause it to fail [70,81]. Recent research, however, suggests that with proper fixation, stability may not be a significant concern [82]. Odate et al. [82] reported that cervical lordosis and segmental height were well maintained after anterior cervical plating. The pseudarthrosis rate and postoperative cervical lordosis of a two-level ACCF were comparable to those of a three-level ACDF, according to Lau et al. [83]. Hirai et al. [84] attempted a hybrid construct composed of ACDF and ACCF, claiming that the hybrid construct improved the biomechanical stability of the ACCF-only construct. In the hybrid group, they performed corpectomy for levels with severe cord compression while discectomy for other levels [84]. In this retrospective study, the hybrid group had no early graft failure, whereas the ACCF group had five cases (16.7%) [84]. Furthermore, the fusion rate in the hybrid group (100%) was significantly higher than in the ACCF group (80.0%) (p=0.02) [84].
Odate et al. [85] in their retrospective analysis assessed the clinical efficacy of ACCF as a revision strategy after poor neurologic improvement following posterior surgery. They discovered that ACCF performed as a revision operation had a higher rate of complications, such as CSF leakage and low neurological improvement, and recommended that ACCF be performed as an initial surgery when poor preoperative prognostic factors for posterior surgery are observed [85]. Niedzielak et al. [86] in their meta-analysis suggested that ACCF with a titanium mesh cage improved clinical outcomes and reduced complication rates. However, only a few studies directly compared the various constructs for ACCF, and this suggestion needs to be validated further [86].
3. Translational osteotomy
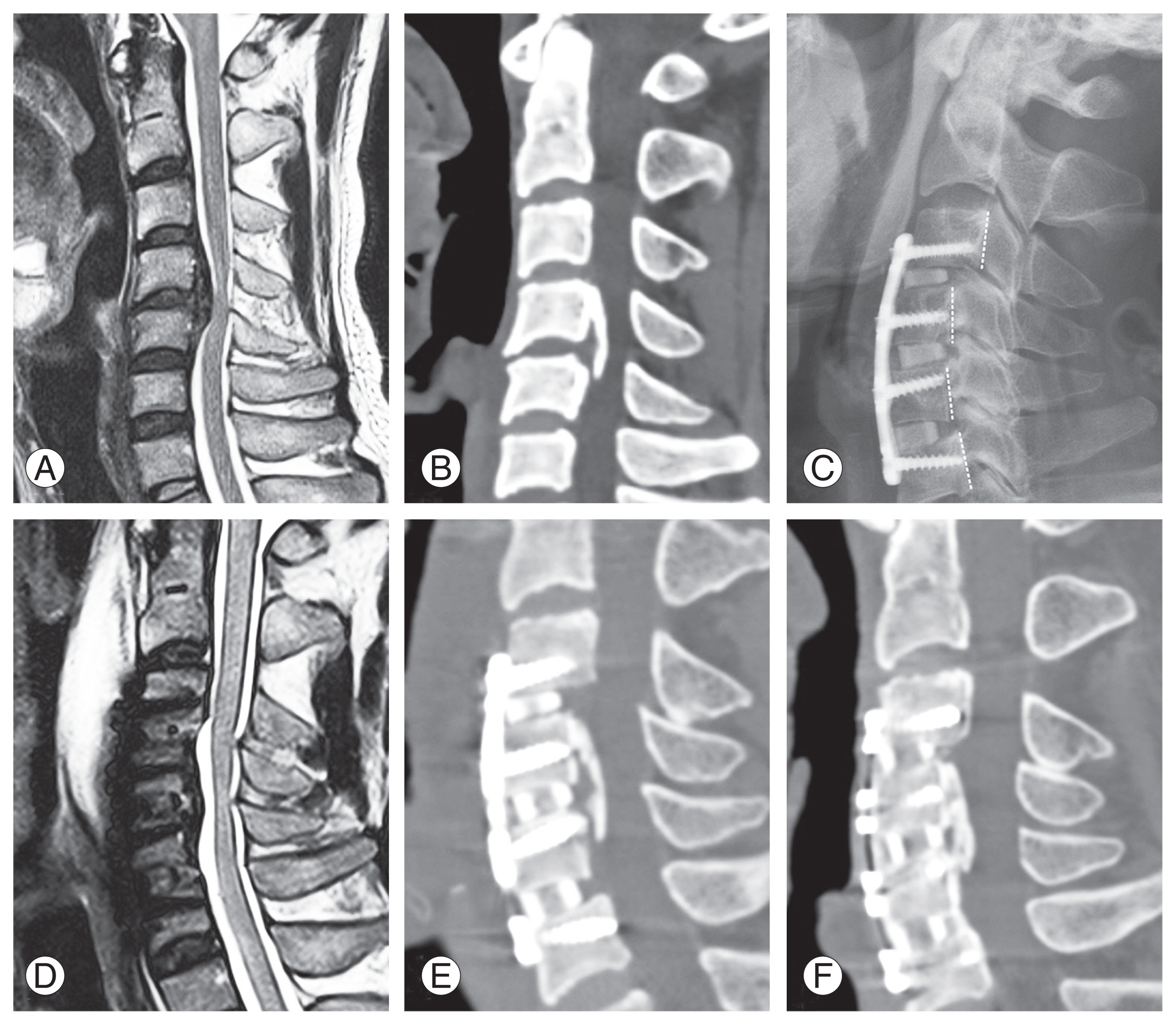
To address the high complication rates associated with ACCF, such as graft dislodgement and CSF leak, a novel anterior translational osteotomy known as “vertebral body sliding osteotomy” (VBSO) has been reported [16]. The concept of VBSO (anterior translation) is similar to that of laminoplasty (posterior translation) in that canal widening is accomplished through bone translation rather than removal. The procedure, which was first reported in the 2014 Cervical Spine Research Society and was published in 2018, involves multi-level discectomy and making bilateral slits at the involved vertebral bodies, allowing the vertebral bodies to float anteriorly [16]. This is followed by the application of an anterior cervical plate and tightening of the screws, allowing for further translation of the vertebral body-OPLL complex anteriorly (Fig. 3) [16,87]. Yang et al. [88] described a similar technique called “anterior controllable ante-displacement fusion” and demonstrated that anterior translational osteotomy could result in favorable outcomes. The indications for VBSO are similar to those for other ADF procedures: (1) K-line (−), (2) rigid kyphosis, and (3) involvement of ≤3 segments [32].

Surgical procedures of vertebral body sliding osteotomy. (A) After performing a complete discectomy, two lateral slits are made at the base of the uncinate process using a high-speed burr. (B) Anterior translation of the vertebral body with ossification of the posterior longitudinal ligament mass with gentle traction. Excessive force should be avoided since this could result in a dural tear or vertebral body injury. (C) While holding the vertebral body in an anteriorly translated position, interbody cages are inserted. The interbody spacer should be tightly fit into the disc space, as a loose fit could result in construct instability. A slight distraction force is applied with a Casper pin distractor to allow control of the vertebral body position. The anterior portion of the vertebral body is removed using a burr. (D) The anterior plate is applied for additional stability. Screw holes should be tapped before insertion since insertion without tapping could push the vertebral body posteriorly. Tightening the screws with the plate could cause additional vertebral body anterior translation.
The main advantage of the VBSO technique is that it eliminates the need to directly manipulate the OPLL mass, lowering the risk of dural tear and CSF leak [89]. In their retrospective comparison of VBSO and ACCF, the authors reported that no dural tear occurred in 24 VBSO patients, whereas it occurred in 10.5% (4/38) of those who underwent ACCF [89]. Furthermore, because it has multiple fixation points with anterior cervical plating, VBSO provides a more stable construct than ACCF resulting in a shorter lever arm and a stronger fixation power [80]. VBSO had a higher 1-year fusion rate than ACDF or ACCF, but the fusion rate at the 2-year follow-up was ambiguous. In addition, VBSO demonstrated significantly lower subsidence than ACCF [80]. It can restore cervical lordosis, as multi-level ACDF is included in the procedure [90]. A retrospective study comparing postoperative sagittal alignment after VBSO and ACCF found that C2–C7 and C0–C2 lordosis as well as C2–C7 sagittal vertical axis, improved significantly after VBSO [90]. Only segmental lordosis at the operative segments, on the other hand, was significantly higher in the VBSO group than in the ACCF group [90].
In terms of neurologic outcomes, the JOA recovery rate of VBSO was comparable to that of ACDF or ACCF, although the OPLL lesion was not directly removed [80]. Moving the ossified mass away from the spinal canal can also produce the effect of direct decompression [32]. Furthermore, VBSO can convert the K-line (−) state into a K-line (+) state due to cervical lordosis restoration and anterior translation of the OPLL lesion [32] (Fig. 4).
Case of vertebral body sliding osteotomy. A 46-year-old male presented with gait disturbance. (A, B) Preoperative magnetic resonance imaging (MRI) and computed tomography (CT) shows significant cord compression by ossification of the posterior longitudinal ligament (OPLL) spanning posterior to C4 and C5 vertebral bodies. Canal occupying ratio was 50%. (C) Vertebral body sliding osteotomy, C4–C5 was performed. Posterior margins of vertebral body signify successful anterior translation of the vertebral bodies. (D, E) Postoperative MRI and CT shows successful cord decompression by anterior translation of the OPLL lesion. (F) Solid fusion is achieved at 1-year postoperative CT.
Conclusions
ADF is beneficial for neurologic recovery, restoring cervical lordosis, alleviating neck pain, and preventing OPLL growth. However, it is associated with a high rate of complications, such as CSF leakage or graft failure. It is recommended for patients who have a high canal occupying ratio, K-line (−), a limited number of levels involved, or significant preoperative neck pain (Table 1). OPLL can be effectively treated with surgical techniques such as ACDF, ACCF, or translational osteotomy. Fig. 5 summarizes suggested surgical strategy selection methods. However, each OPLL cervical myelopathy has unique characteristics, and a single guideline cannot be applied universally. The surgical method should be carefully chosen after taking into account the benefits and limitations of ADF as well as patient-specific factors.
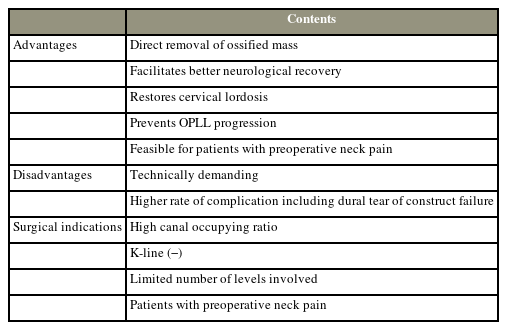
Summary of advantages, limitations, and indications of anterior decompression and fusion for the treatment of cervical myelopathy caused by OPLL
Acknowledgments
This review manuscript is being submitted in place of “Significance of vertebral body sliding osteotomy for the treatment of cervical ossification of the posterior longitudinal ligament” which is the winning paper of 2021 APSS-ASJ Best Paper Award.
Notes
No potential conflict of interest relevant to this article was reported.
Author Contributions
Conception and design: Sehan Park, Dong-ho Lee; drafting of the manuscript: Sehan Park; critical revision: Choon Sung Lee, Chang-Ju Hwang, Jae Jun Yang, Jae Hwan Cho; and final approval of the manuscript: all authors.

